CONTENTS
- Basics
- Epidemiology & causes of organizing pneumonia
- Presentation
- Imaging findings
- Laboratory studies
- Invasive diagnostics
- Differential diagnosis
- Management
- Questions & discussion
Organizing pneumonia (OP) is a pathological form of lung response to various injuries. It involves the formation of organized plugs of fibrous tissue within alveoli. Typically, this inflammation is responsive to steroid.

SOP vs. COP
- Secondary organizing pneumonia (SOP) refers to organizing pneumonia caused by a known stimulus (e.g., infection, medication).
- Cryptogenic organizing pneumonia (COP) refers to organizing pneumonia without any known triggering factor. In reality COP isn't a single distinct entity, but rather COP probably includes a variety of conditions involving organizing pneumonia triggered by various stimuli that we are unable to detect (e.g., undiagnosed viral infections).
- OP (organizing pneumonia) encompasses both SOP and COP. OP might the best way to conceptualize COP and SOP because:
- The treatment of COP and SOP is similar (aside from avoiding and treating known triggers in SOP).
- The radiology of COP and SOP is largely identical.
- COP was previously known as BOOP (bronchiolitis obliterans organizing pneumonia). BOOP led to some confusion, because bronchiolitis obliterans is a distinct disorder entirely. BOOP was a glorious term, but it has unfortunately been abandoned in current parlance.
microscopic areas of organizing pneumonia
- Microscopic foci of organizing pneumonia may be seen in the context of various other predominant pathologies (e.g., vasculitis, lung cancer, lymphoma, eosinophilic pneumonia, diffuse alveolar damage, or nonspecific interstitial pneumonia).(24559802)
- Tiny areas of organizing pneumonia on histology don't establish a clinical diagnosis of organizing pneumonia. For the remainder of this chapter, patients with microscopic foci of organizing pneumonia in the context of another predominant disorder will not be regarded as having a clinico-radiological diagnosis of organizing pneumonia (OP).
Organizing pneumonia may be triggered by a broad range of lung insults. The evidentiary support behind many of these triggers is circumstantial, making it impossible to construct any definitive list. Nonetheless, having some list is useful – because these factors should always be considered prior to concluding that the patient's organizing pneumonia is cryptogenic (COP). Prominent causes of OP include the following: (35294814)
cryptogenic organizing pneumonia (COP)
- COP is quite rare, with most referral centers seeing no more than a few cases per year.
- COP can occur at any age, but might be more common in somewhat older patients (with a mean age of onset of 58 years old).(Fishman 2023)
secondary organizing pneumonia (SOP)
infection
- Bacteria (especially atypical organisms):
- Burkholderia cepacia.
- Chlamydia pneumoniae.
- Coxiella burnetii.
- Legionella pneumophila.
- Mycoplasma pneumoniae.
- Nocardia asteroides.
- Pseudomonas aeruginosa.
- Serratia marcescens.
- Staphylococcus aureus.
- Streptococcus pneumoniae.
- Viruses:
- Adenovirus.
- COVID-19.
- CMV (cytomegalovirus).
- HHV-7 (human herpesvirus 7)
- HIV.
- HSV (herpes simplex virus).
- Influenza virus.
- Parainfluenza virus.
- RSV (respiratory syncytial virus).
- Parasites:
- Plasmodium vivax.
- Dirofilaria immitis.
- Fungi:
- Aspergillus.
- Cryptococcus neoformans.
- Penicillium janthinellum.
- Pneumocystis.
- CVID (common variable immunodeficiency). However, OP due to CVID is probably most accurately regarded as GLILD (granulomatous and lymphocytic interstitial lung disease), which is discussed further here: 📖
drugs and exposures
- Medications most often implicated (complete list at PneumoTox.com 📖):
- Amiodarone, dronedarone.
- Benralizumab.
- Cancer therapies:
- Chemotherapy: Azacitidine, Bleomycin, Bortezomib, Busulfan, cyclophosphamide, doxorubicin, etoposide, FOLFIRI, FOLFOX, fludarabine, hydroxyurea, leflunomide, melphalan, methotrexate, oxaliplatin, pemetrexed, temozolomide.
- Targeted therapies: Ceritinib, Cetuximab, crizotinib, dasatinib, erlotinib, imatinib,
- Checkpoint inhibitors.
- Carbamazepine.
- Clopidogrel.
- Dabigatran.
- Daptomycin.
- Hydralazine.
- Interferon alpha/beta.
- Mesalamine.
- Nitrofurantoin.
- Phenytoin.
- Propylthiouracil.
- Risperidone.
- Rituximab.
- Sertraline.
- Sirolimus.
- Statins.
- Sulfasalazine.
- TNF-inhibitors (adalimumab, etanercept).
- Inhalation exposures:
- Freebased cocaine.
- Synthetic marijuana.
- Aerosolized textile dye (e.g., Acramin FWN).
- Mustard gas.
- EVALI (electronic cigarettes and vaping-associated lung injury). (35038455)
- Aspiration.
malignancy
- Radiotherapy for thoracic malignancy (especially 3-6 months after radiation for breast cancer).(Fishman 2023)
- Chemotherapy (see medications above).
- Hematologic malignancy: leukemia, lymphoma.
- Allogeneic hematopoietic stem cell transplantation.
rheumatologic/inflammatory disorders
- Disorders associated with OP:
- Rheumatoid arthritis.
- Polymyositis or dermatomyositis (including anti-synthetase syndrome).
- Sjogren syndrome.
- Systemic sclerosis.
- Lupus.
- Inflammatory bowel disease.
- Vasculitis, especially:
- GPA (granulomatosis with polyangiitis).
- EGPA (eosinophilic granulomatosis with polyangiitis).
- OP may occur in the context of a known inflammatory disorder. Alternatively, patients with OP may present weeks to months prior to developing an associated connective-tissue disorder.(35294814)

OP usually presents clinically as a chronic pneumonia
- Trigger:
- Antecedent respiratory tract infection (upper or lower) may be reported in about a third of patients with COP.
- Patients with SOP have exposure to various other triggers.
- Subacute symptoms develop over a period of weeks to months. These often include:(35294814)
- Dry cough.
- Progressive dyspnea.
- Constitutional symptoms may be prominent:
- Fever.
- Malaise.
- Weight loss, anorexia.
- Patients are often treated initially with antibiotics for a presumed bacterial pneumonia. Often the diagnosis of OP is only recognized only after failure to respond to a few rounds of antibiotics.
physical examination
- Examination is often normal.
- Crackles are often present.
- Clubbing is almost always absent.(35038455)
Imaging findings in OP are diverse and often nonspecific. However, there are some patterns which may be recognizable – potentially suggesting the diagnosis. Migration of opacities over time is especially suggestive of OP. COP (cryptogenic organizing pneumonia) and SOP (secondary organizing pneumonia) generally have the same radiologic appearance. (34246383)
(#1) consolidation
Consolidation is the most common finding, seen in ~85% of patients.
distribution
- Lower lobe predominance is often seen.
- Axial distribution:
- (1) Subpleural distribution is most commonly seen. This may resemble the pattern seen in CEP (chronic eosinophilic pneumonia).
- (2) Peribronchovascular distribution may occur, sometimes with thickening of the adjacent airway.
- (3) Subpleural and peribronchovascular distribution may occur together.
- Migration of lesions over time may be seen with serial imaging.
- Band-like distribution.
appearance
- Often poorly marginated.
- Size varies.
- Consolidation contains air bronchograms and may contain bubbly lucencies (areas of unaffected alveoli).
- Bronchial wall thickening and dilation may occur.(28106480)
- 1-10 mm centrilobular nodular opacities may occur, usually combined with consolidation. If seen, these may help support a diagnosis of OP (as opposed to chronic eosinophilic pneumonia).(Walker 2019)

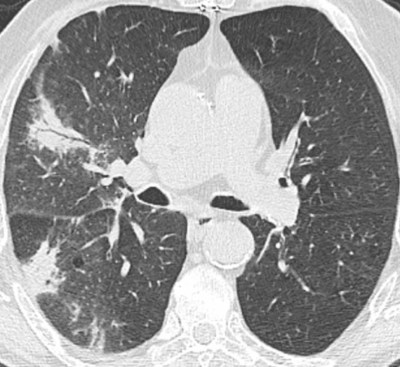
(#2) nodule or mass
nodular COP
- ~2-8 large nodular lesions may occur, usually as part of a mixed pattern.(28106480)
- The size often ranges from ~8 mm to 5 cm.(Walker 2019)
- Clues to the presence of COP may include:
- Air bronchograms may be present within nodules.
- Irregular margins and/or broad pleural tags may be seen. In some cases, a halo sign may be present, mimicking angioinvasive aspergillosis. (24559802)
- Bronchovascular distribution may occur.
solitary nodule/mass (focal organizing pneumonia)
- Patients may present with a single nodule/mass that simulates malignancy.
- The lesion is often in an upper lobe.
- Patients are usually asymptomatic or mildly symptomatic (e.g., constitutional symptoms, dry cough, dyspnea, chest pain). (35038455)
- Most cases are cryptogenic. (29515668)

(#3) ground glass opacity (GGO), in various configurations
ground glass opacities in COP
- Ground glass opacities are present in ~75% of patients:
- These are usually part of a mixed pattern (e.g., in association with consolidation).
- Occasionally ground glass opacities are the primary manifestation of COP.
- Characteristic of ground glass opacities:
- GGO is often bordered by thin areas of consolidation (reverse-halo sign; discussed below).
- A crazy-paving pattern (due to septal thickening) can occur.(35038455)
- Distribution:
- The overall distribution of GGO is usually bilateral and random.
- Central disease with subpleural sparing can occur. In about half of patients with bilateral lower lobe opacities and subpleural sparing suggestive of NSIP (nonspecific interstitial pneumonia), the surgical lung biopsy actually reveals organizing pneumonia.(34246383)
- Lobular sparing may also occur, wherein small areas of lung appear normal.(37289644)


reverse halo (atoll) sign
- This consists of a focal rounded area of ground glass opacity that is surrounded by a more or less complete ring of consolidation. 📖 (28106480)
- The reverse-halo sign is seen in only ~20% of patients with COP, but may be suggestive of the diagnosis.
- In general, the edges of lung opacities in COP are often wispy.(Shepard 2019)
- Further discussion of the reverse halo (atoll) sign here: 📖

(#4) perilobular opacities
Further discussion of perilobular patterns here: 📖


diffuse small centrilobular nodules
- This is a very rare manifestation of OP.
- Diffuse centrilobular nodules may result from an inhalational exposure that injures the bronchioles, such as:
- Synthetic marijuana.
- Some patients with EVALI (E-cigarette and Vaping-induced acute lung injury). (37289644)
pertinent negatives
- The following findings would argue against a diagnosis of cryptogenic organizing pneumonia (COP):
- Honeycombing.
- Substantial pleural involvement (e.g., pleural effusion or pleural thickening). Pleural effusion may be seen in ~5% of patients, but this is usually small and unilateral.
- Prominent lymphadenopathy. (35294814) Mediastinal lymphadenopathy may occur in about a third of patients, but usually only 1-2 nodal stations are involved and enlargement is mild. (29515668)
- Cavitation.
- ⚠️ Note, however, that patients with secondary organizing pneumonia may exhibit a combination of organizing pneumonia plus another disorder.
laboratory findings in OP are nonspecific
- CRP (C-reactive protein) is elevated in most patients (e.g., mean of 97 mg/L and interquartile range of 46-150 mg/L). (31120857)
- CRP may help predict relapses while tapering steroid.(35038455)
- Procalcitonin was <0.28 ng/ml in 89% of patients with COP within one series.(31120857) Procalcitonin is far from perfect, but a low procalcitonin value may help call the diagnosis of typical community-acquired pneumonia into question. The combination of a low procalcitonin plus an elevated CRP level may help point to a non-bacterial inflammatory process.
investigation should be directed towards exclusion of other diagnoses
- Laboratory studies are aimed at evaluation for alternative or underlying diagnoses.
- For patients with a pneumonia-like presentation, the workup should include the evaluation of community-acquired pneumonia: 📖
- OP is often due to an underlying rheumatologic process, so laboratory studies should be used to evaluate this possibility. For most patients, the following may be reasonable: ANA, RF, anticentromere antibody, anti-SCL70, and a myositis panel.
Organizing pneumonia may represent a component of another pathology, so simply finding a small area of organizing pneumonia doesn't establish the diagnosis of COP.
bronchoscopy
role of bronchoscopy in diagnosis
- Bronchoscopy may be useful to exclude alternative diagnoses, most frequently:
- Infection.
- Eosinophilic pneumonias.
- Bronchoscopy cannot confirm the diagnosis of OP with certainty. Even if transbronchial biopsies are performed, these aren't generally large enough to fully clarify the histology. Small areas of OP may occur with a variety of disease processes, so this finding alone on a small biopsy doesn't necessarily prove the primary diagnosis as OP. If bronchoscopic biopsy is used to reach a working diagnosis of OP, follow-up imaging is important to establish resolution. (37289644)
BAL (bronchoalveolar lavage) usually shows a pattern of mixed cellularity
- Lymphocytes, neutrophils, and eosinophils are usually elevated compared to normal:
- Lymphocytes are often ~20-40% (normally <15%).
- Neutrophils are often ~5-15% (normally <3%).
- Eosinophils are often ~5% and usually less frequent than lymphocytes (normally eosinophils are <1%).
- This pattern of mixed cellularity may be seen with some other diseases as well: 📖
- Foamy macrophages may be observed. (32194763)
transthoracic needle biopsy
- The yield of a transthoracic needle biopsy for the diagnosis of organizing pneumonia is unknown. However, this could be a reasonable consideration in some patients, for example:
- Dense area of consolidation is present.
- Consolidation is peripherally located (especially abutting the pleura).
surgical biopsy
- Definite diagnosis generally requires a surgical biopsy, but this isn't always clinically wise.
- If the diagnosis is very unclear and the patient can tolerate a surgical biopsy, this may be advisable. Alternatively, if the patient is unstable and/or available data is extremely suggestive of COP, then a trial of steroid may be reasonable.
COP has a variety of different presentations, so it's impossible to construct a concise list of disorders that may be confused with COP. Some pertinent differential alternative diagnoses may include the following:
- Differential diagnosis of pneumonia. 📖
- Differential diagnosis of pneumonia that fails to respond to antibiotics. 📖
- Differential diagnosis of migratory infiltrates. 📖
Treatment of SOP and COP are the same, with the exception that treatment of SOP additionally involves management of any cause. (35038455)
treatment/avoidance of any potential causes of organizing pneumonia
- Always consider the list of possible causes of organizing pneumonia: 📖.
- In particular, any medications potentially associated with organizing pneumonia should be discontinued.
steroid is the front line therapy
- Steroid is the cornerstone of initial therapy. Steroid usually causes dramatic clinical improvement within days, with radiographic clearing following within a couple months.
- A commonly utilized steroid regimen is as follows:
- Start with 4 weeks of prednisone at 0.5-1 mg/kg/day (up to a maximal dose of 60 mg/day). If the patient responds favorably, the dose may be dropped to 0.25 mg/kg/day and subsequently slowly weaned off over six months. (34246383)
- However, some authors recommend a three-month steroid taper to minimize steroid exposure. The duration of steroid therapy may not actually affect the risk of relapse. (Fishman 2023)
- However, not every patient requires this regimen of steroid:
- Mild or asymptomatic disease may not require treatment. (Fishman 2023)
- Steroid dose may be titrated based on the disease severity. For a patient with moderate severity illness, it may be reasonable to start with a lower dose of steroid (e.g., 20 mg/day prednisone). (32194763) For patients with severe hypoxemic respiratory failure, higher initial steroid doses are reasonable.
nonresponse to steroid is uncommon
- Failure to respond to steroid is unusual, but might occur in ~20% of patients.
- Important considerations:
- (1) Is OP the correct diagnosis?
- (2) Is there an unrecognized cause of OP which is causing ongoing lung injury?
- Additional treatment options may include cyclophosphamide, azathioprine, mycophenolate, or rituximab. (35038455) There isn't high level evidence or wide experience with these therapies.
relapse is common
- Relapse commonly occurs with tapering off steroid.
- Fortunately, relapse generally responds to therapy (e.g., higher doses of steroid), so that relapse doesn't affect overall outcomes substantially.(34246383)
- Monitoring for relapse may include:
- Clinical symptoms.
- Radiographic follow-up (with serial chest films).
- Following inflammatory markers such as CRP (C-reactive protein).(35038455)
- Occasional patients may require a steroid-sparing agent to facilitate weaning off steroid (e.g., azathioprine). (34246383)
- OP usually resolves nicely with steroid therapy.
- OP may progress to fibrosis that resembles fibrotic NSIP (nonspecific interstitial pneumonia). (31200868) Such patients might actually have an overlap of OP and NSIP (e.g., due to an underlying connective tissue disorder).
To keep this page small and fast, questions & discussion about this post can be found on another page here.
Guide to emoji hyperlinks 
= Link to online calculator.
= Link to Medscape monograph about a drug.
= Link to IBCC section about a drug.
= Link to IBCC section covering that topic.
= Link to FOAMed site with related information.
- 📄 = Link to open-access journal article.
= Link to supplemental media.
References
- 24559802 Baque-Juston M, Pellegrin A, Leroy S, Marquette CH, Padovani B. Organizing pneumonia: what is it? A conceptual approach and pictorial review. Diagn Interv Imaging. 2014 Sep;95(9):771-7. doi: 10.1016/j.diii.2014.01.004 [PubMed]
- 28106480 Zare Mehrjardi M, Kahkouee S, Pourabdollah M. Radio-pathological correlation of organizing pneumonia (OP): a pictorial review. Br J Radiol. 2017 Mar;90(1071):20160723. doi: 10.1259/bjr.20160723 [PubMed]
- 29515668 Van de Kerkhove C, De Wever W, Verbeken EK, Deroose C, Nackaerts K. An unusual presentation of a more common disease entity. Breathe (Sheff). 2018 Mar;14(1):49-53. doi: 10.1183/20734735.013417 [PubMed]
- 31120857 Ito A, Ishida T, Tachibana H, Arita M, Yamazaki A, Washio Y. Utility of procalcitonin for differentiating cryptogenic organising pneumonia from community-acquired pneumonia. Clin Chem Lab Med. 2019 Sep 25;57(10):1632-1637. doi: 10.1515/cclm-2019-0175 [PubMed]
- 32194763 Afthinos A, Antonakis E, Horti M, Markatis E, Pagratis K, Papanikolaou IC. An unusual disease with an interesting sign. Breathe (Sheff). 2020 Mar;16(1):190302. doi: 10.1183/20734735.0302-2019 [PubMed]
- 33280768 Geier M, Picart G, Khatchatourian L, Samaison L, Robinet G, Couturaud F, Bizien N. A 60-Year-Old Woman With Recurrent Hemoptysis. Chest. 2020 Dec;158(6):e273-e277. doi: 10.1016/j.chest.2020.06.069 [PubMed]
- 34246383 Chong WH, Saha B, Ibrahim A, Smith TC. Dyspnea in a 57-Year-Old Man With Recent Viral Illness. Chest. 2021 Jul;160(1):e19-e24. doi: 10.1016/j.chest.2021.01.012 [PubMed]
- 34407978 Raghu G, Meyer KC. Cryptogenic organising pneumonia: current understanding of an enigmatic lung disease. Eur Respir Rev. 2021 Aug 17;30(161):210094. doi: 10.1183/16000617.0094-2021 [PubMed]
- 35038455 Cherian SV, Patel D, Machnicki S, Naidich D, Stover D, Travis WD, Brown KK, Naidich JJ, Mahajan A, Esposito M, Mina B, Lakticova V, Cohen SL, Muller NL, Schulner J, Shah R, Raoof S. Algorithmic Approach to the Diagnosis of Organizing Pneumonia: A Correlation of Clinical, Radiologic, and Pathologic Features. Chest. 2022 Jul;162(1):156-178. doi: 10.1016/j.chest.2021.12.659 [PubMed]
- 35294814 King TE Jr, Lee JS. Cryptogenic Organizing Pneumonia. N Engl J Med. 2022 Mar 17;386(11):1058-1069. doi: 10.1056/NEJMra2116777 [PubMed]
- 37289644 Marquis KM, Hammer MM, Steinbrecher K, Henry TS, Lin CY, Shifren A, Raptis CA. CT Approach to Lung Injury. Radiographics. 2023 Jul;43(7):e220176. doi: 10.1148/rg.220176 [PubMed]
Books:
- Shah, P. L., Herth, F. J., Lee, G., & Criner, G. J. (2018). Essentials of Clinical pulmonology. In CRC Press eBooks. https://doi.org/10.1201/9781315113807
- Shepard, JO. (2019). Thoracic Imaging The Requisites (Requisites in Radiology) (3rd ed.). Elsevier.
- Walker C & Chung JH (2019). Muller’s Imaging of the Chest: Expert Radiology Series. Elsevier.
- Palange, P., & Rohde, G. (2019). ERS Handbook of Respiratory Medicine. European Respiratory Society.
- Rosado-De-Christenson, M. L., Facr, M. L. R. M., & Martínez-Jiménez, S. (2021). Diagnostic imaging: chest. Elsevier.
- Murray & Nadel: Broaddus, V. C., Ernst, J. D., MD, King, T. E., Jr, Lazarus, S. C., Sarmiento, K. F., Schnapp, L. M., Stapleton, R. D., & Gotway, M. B. (2021). Murray & Nadel’s Textbook of Respiratory Medicine, 2-Volume set. Elsevier.
- Fishman's: Grippi, M., Antin-Ozerkis, D. E., Cruz, C. D. S., Kotloff, R., Kotton, C. N., & Pack, A. (2023). Fishman’s Pulmonary Diseases and Disorders, Sixth Edition (6th ed.). McGraw Hill / Medical.

