
With the publication of the SOLIDARITY trial, there is increasing consensus that remdesivir doesn’t affect mortality. However, there continues to be debate about whether remdesivir affects hospital length of stay. Let’s dig into the data here to try to reconcile differences between ACTT-1 and SOLIDARITY.
Did ACTT-1 actually show that remdesivir reduces the hospital length of stay? Three major limitations.
limitation #1: “time to recovery” versus “length of stay”
You might think that remdesivir decreased hospital length of stay in the ACTT-1 trial, because that’s what most of the press suggested. But that’s not necessarily true.
The primary endpoint in ACTT-1 was “time to recovery” which was defined as the first point in time when the patient was either discharged or considered fit for discharge (e.g. awaiting discharge due to infection-control purposes). The investigators chose this endpoint because patients with COVID may often require ongoing hospital admission after they recover, while arrangements are being made to safely discharge them without infecting others.
There is a subtle, but critical difference between these two endpoints:
- Time to recovery is the best endpoint to determine if remdesivir is having any minimal effect on the disease course. It may help answer the question: is remdesivir having any positive effect at all on the patient?
- Length of stay is the best endpoint to determine if remdesivir has any pragmatic effect on the patient’s duration of hospitalization. If you’re trying to see how many beds remdesivir will free up, length of stay is the most important endpoint.
Remdesivir did cause a statistically significant reduction in the time to recovery (more on this below). Remdesivir also did decrease the median hospital length of stay (12 days vs. 17 days) – but there is no data included regarding whether this latter endpoint was statistically significant. The length of stay endpoint is glossed over in the manuscript, suggesting that it wasn’t as impressive as the time to recovery endpoint.
limitation #2: Wide confidence intervals
ACTT-1 found that remdesivir increased the rate ratio for recovery by a factor of 1.29, with a 95% confidence interval of 1.12-1.49. However, there were some baseline imbalances in the data (patients in the control group tended to be sicker). After attempting to correct for these imbalances, remdesivir increased the rate ratio for recovery by a factor of 1.26, with a 95% confidence interval of 1.09-1.46.
Those are relatively broad confidence intervals. A rate ratio of 1.46 could be quite beneficial, whereas a rate ratio of 1.09 might not be clinically meaningful.
limitation #3: Double-blind RCT vs real-world length of stay
A double-blind RCT is the best way to determine whether a drug has any physiological effect. It eliminates biases and prescribing patterns, in order to isolate the true biological effect of the drug.
However, a double-blind RCT might not the best way to evaluate hospital length of stay! The act of prescribing remdesivir itself may extend the hospital length of stay (for example, if the patient is kept in the hospital solely to finish their courses of remdesivir). A RCT will, by definition, be incapable of capturing this effect. This may cause the results of the RCT to fail to generalize into clinical reality.
conclusions on ACTT-1
It was widely perceived that ACTT-1 proved that Remdesivir shortens hospital length of stay by five days. However, this just isn’t what the data shows.
This study leaves it quite murky what effect remdesivir would actually have on real-world clinical practice. First, the effect of remdesivir on time to recovery has a very broad 95% confidence interval. Second, it’s dubious whether the time to recovery will translate directly into the length of stay. Finally, it’s possible that the act of prescribing remdesivir will itself prolong the hospital length of stay. All told, ACTT-1 fails to provide robust information about how remdesivir will affect real-world hospital length of stay.
SOLIDARITY: Remdesivir actually prolonged hospital length of stay!
The SOLIDARITY trial is a massive, multi-center, open-label, pragmatic trial. Pragmatic trials are often demeaned in the scientific literature as less “pure” than placebo-controlled RCTs. However, pragmatic trials simply answer a different question:
- Pragmatic trials evaluate the effects that the treatment has in the context of real-world clinical practice. This includes the physiological effect of the medication as well as other unforeseen effects that the medication may have on the patient’s care (e.g. extending the hospital length of stay to finish the remdesivir course). This is less “clean” than a placebo-controlled trial, but provides a closer gauge of clinical reality.
- Double-blind placebo RCTs seek to isolate the effect of the medication on the patient’s physiology. This focuses on a more limited question of whether the medication works biologically – not necessarily how the medication works in the context of the larger healthcare system.
One advantage of pragmatic trials is that they can enroll more patients. For example, the SOLIDARITY trial evaluated remdesivir in 5451 patients (compared to 1062 patients in ACTT-1).
The SOLIDARITY trial shows that remdesivir actually increases the hospital length of stay slightly:

Why? Patients in all three open-label treatment arms (remdesivir, hydroxychloroquine, and lopinavir) had increased lengths of stay by ~1-3 days, because some patients were kept in the hospital to finish their medication course. No differences in length of stay were observed between these three medications, suggesting that they all were equally lacking in efficacy.
Essentially, ACTT-1 demonstrated that remdesivir might work in a magical universe (where patients were discharged immediately when they met discharge criteria). SOLIDARY showed that in the messy reality of clinical medicine, remdesivir doesn’t actually help – and it may actually prolong hospitalization.
where does this leave us with remdesivir?
Remdesivir doesn’t improve mortality, intubation, or hospital length-of-stay. Consequently, the World Helath Organization has recommended against the use of remdesivir.
In the United States, enthusiasm for remdesivir will likely persist. Indeed, given ongoing enthusiasm for hydroxychloroquine, it may be impossible to ever suppress enthusiasm for remdesivir.
This is a complex topic, because it remains possible that remdesivir does cause a minimal clinical improvement (which is likely clinically insignificant). Current studies cannot refute with 100% certainty that remdesivir causes some non-zero benefit (that is statistically impossible, as it would require an infinite number of patients).
Consequently, clinicians will be left to weight two possibilities:
- Benefit: Remdesivir might cause a minimal clinical benefit (one which is too small to be consistently observed in available trials).
- Harm: Remdesivir remains an experimental drug which isn’t FDA approved for any indication. It is known to cause kidney failure and hepatitis. It’s almost certain to cause other side-effects which we haven’t yet discovered. So remdesivir remains a wild-card, which could cause unknown harm.
In areas of the United States which are flush with remdesivir, it may be reasonable to use it among patients with no contraindications (e.g. excellent renal function, no pregnancy, normal liver function). However, for most patients the risk and cost of this drug may be challenging to justify. Hopefully further RCTs will help elucidate the balance of risk vs. harm.
prior posts on remdesivir
- The original compassionate-use study
- What a dumpster fire (PulmCrit)
- Wang et al., first RCT on remdesivir
- Discussion including viral kinetics (PulmCrit)
- Two more trials just published on Remdesivir (RebelEM, Salim Rezaie)
- Remdesivir – The first real trial (First10EM, Justin Morgenstern)
- 5 day vs. 10-day trial
- Discussion including some signs of toxicity (PulmCrit)
- Remdesivir RCT #3 (5d vs. 10d) (RebelEM, Salim Rezaie)
- ACTT-1
- Discussion of preliminary results (PulmCrit)
- ACTT-1 (TheBottomLine, by Fraser Magee)
- Case of the Partial Cohort (EMNerd, Rory Spiegel)
- ACTT-1 Trial (First10EM, Justin Morgenstern)
- Adaptive COVID-19 Treatment Trial part I (RebelEM, Salim Rezaie)
Image credits: Photo by Aron Visuals on Unsplash
- Pulmcrit wee: The cutoff razor - April 15, 2024
- PulmCrit Blogitorial – Use of ECGs for management of (sub)massive PE - March 24, 2024
- PulmCrit Wee: Propofol induced eyelid opening apraxia – the struggle is real - March 20, 2024
Great article. I would point out that in the real world, if I have a patient that is needing oxygen, get’s started on remdesivir and then improves off oxygen within two days I have no problem discharging them. Maybe remdesivir did help reduce LOS in that case and save the hospital some money?
The problem then becomes, was this patient really sick enough to need it or not and would have gotten better just with the dex I gave, or if I hadn’t given it, would they have recovered as quickly?
I agree that remdesivir effect is tiny if any at all, but I work at a large quaternary hospital where it is common practice now to discharge patients whenever they are ready with no need to finish remdesivir course, so that could be a point for Gilead to say that their endpoint would actually translate to a real world benefit. On the other hand I think a detail that hasn’t been really considered is that on Solidarity 47% of the remdesivir arm (control and intervention) received corticosteroids which is definitely more effective than remdesivir, while on ACTT1 it was only… Read more »
Here are clinical trials in which remdesivir failed to shorten hospital stays or reduce deaths:


https://www.medrxiv.org/content/10.1101/2020.10.15.20209817v1
https://pubmed.ncbi.nlm.nih.gov/33264556/
https://rebelem.com/remdesivir-in-moderate-covid-19/
https://www.medrxiv.org/content/10.1101/2020.08.10.20171637v1
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7190303/
https://finance.yahoo.com/chart/GILD
Remdesivir is totally useless against all viral diseases, including the flu, common cold, and Ebola:

https://www.nejm.org/doi/full/10.1056/NEJMoa1910993
Remdesivir is fraudulently indicated for end-stage COVID-19 when in truth, it has absolutely no benefit in reducing the cytokine storm, in fact, might make it slightly worse:
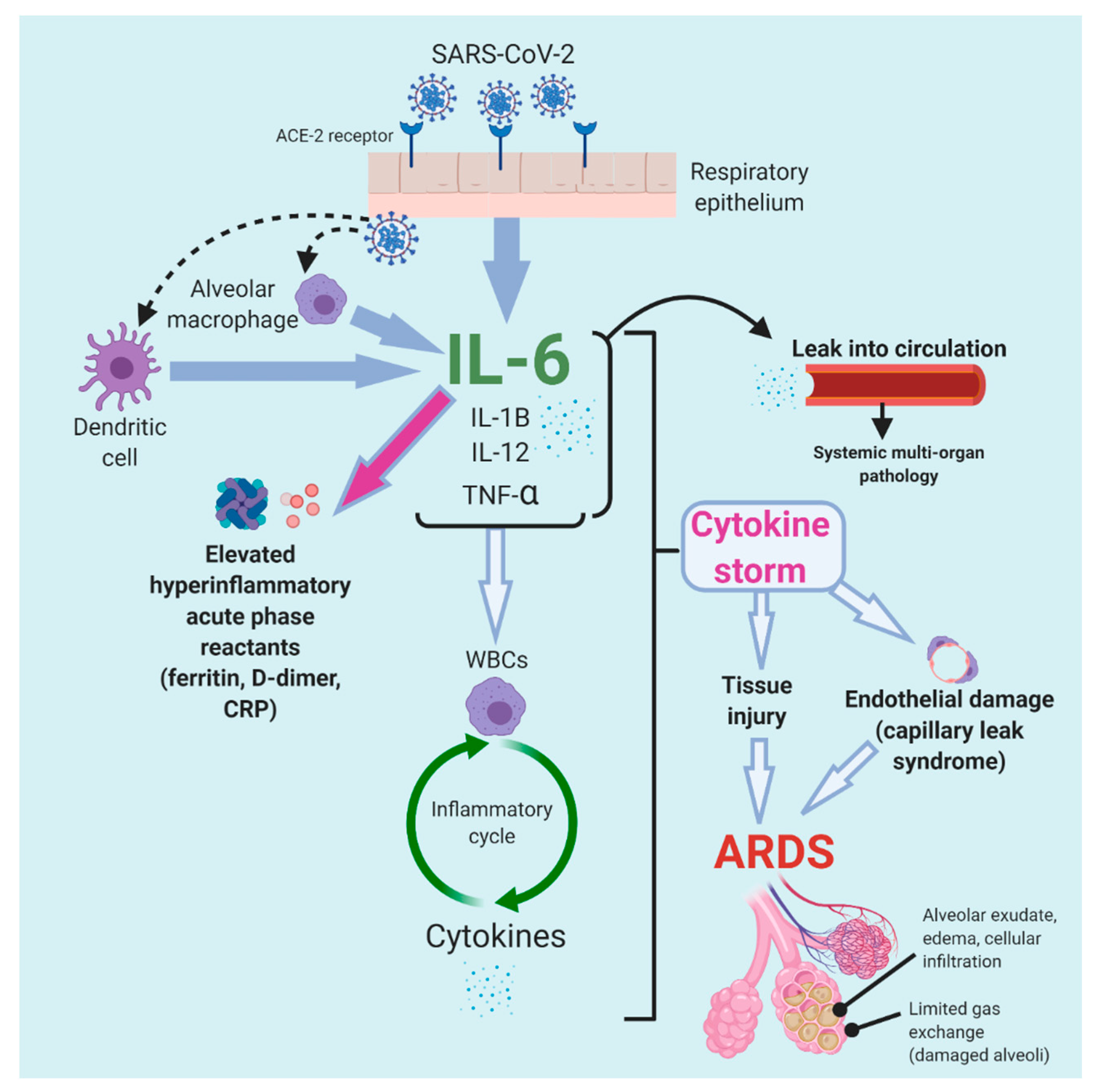
https://www.nature.com/articles/s41591-020-1051-9
https://www.nature.com/articles/s41591-020-1051-9/figures/5
Remdesivir, hydroxychloroquine, and chloroquine cause cardiovascular QT interval prolongation and sinus bradycardia:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7571425/
https://www.sciencedirect.com/science/article/abs/pii/S0022073620305835
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7598346/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7682945/
Remdesivir causes liver damage:
https://theprint.in/health/govt-reviewing-remdesivir-use-for-covid-after-hospitals-report-liver-damage-in-patients/454169/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7381904/
https://pubmed.ncbi.nlm.nih.gov/33006138/
Remdesivir causes kidney damage:
https://www.rxlist.com/consumer_remdesivir_rdv/drugs-condition.htm
https://pubmed.ncbi.nlm.nih.gov/33252992/
As an intensivist I am not starting it in ICU, only continuing if already started on ward. So many patients have AKI or elevated LFT and no study showing benefit in critical care. Some patients push back to get the “antiviral “…