Introduction
This is one of my favorite diagnoses. When first encountered, it may seem bewildering and difficult to treat (1). Indeed, standard ACLS algorithms may fail with these patients. However, once understood, this disorder is easily treated and patients typically improve rapidly.
Pathophysiology & definition
BRASH syndrome is defined as a combination of the following:
- Bradycardia
- Renal failure
- AV node blocker: beta-blocker, verapamil, or diltiazem (2)
- Shock
- Hyperkalemia
This syndrome is due to a vicious cycle in the setting of medications, hyperkalemia, and renal failure (shown below). Renal failure causes hyperkalemia and may cause the accumulation of some AV node blockers (e.g. atenolol, nadolol). Hyperkalemia synergizes with AV node blockers to cause bradycardia and hypoperfusion. Hypoperfusion, in turn, causes worsening of the renal failure.
The pathophysiologic key of BRASH syndrome is the ability of hyperkalemia to synergize with AV node blockers to cause bradycardia (Hegazi 2012, Letavernier 2006, Bonvini 2006). This has been proven using a canine model (Nugent 1984, Jolly 1991).
Evidence & parsing out BRASH as a specific diagnostic entity
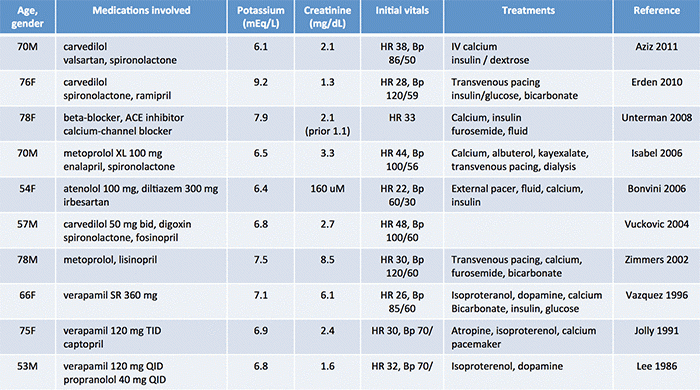
Several authors have reported these cases (table below)(3). Nonetheless, this entity remains under-recognized. BRASH syndrome is often blamed entirely on hyperkalemia. Since hyperkalemia triggers a “panic value” call to the provider, it is easy to anchor onto this diagnosis. Meanwhile, the presence of AV node blockers lurking in the background may seem less important.
 BRASH syndrome represents an overlap between hyperkalemia and AV node blocker intoxication. Some features may help distinguish these entities:
BRASH syndrome represents an overlap between hyperkalemia and AV node blocker intoxication. Some features may help distinguish these entities:
Differentiating BRASH vs. pure hyperkalemia:
- Degree of hyperkalemia: Patients with BRASH syndrome may have only mild hyperkalemia, with bradycardia due to synergy between hyperkalemia and the AV node blocker. In contrast, for hyperkalemia alone to cause bradycardia usually requires a more dramatic elevation of the potassium level (4).
- EKG with disproportionate bradycardia: The sequence of appearance of various EKG findings in hyperkalemia can be variable. Nonetheless, bradycardia without other EKG features of hyperkalemia (e.g. QRS widening, peaked T-waves) may favor BRASH syndrome (Lee 1986).

Differentiating BRASH vs. pure AV node blocker intoxication:
- Presence of hyperkalemia: Patients with beta-blocker or calcium channel blocker intoxication may or may not have hyperkalemia, whereas this is an essential component of BRASH syndrome.
- History: Patients with significant AV blocker intoxication usually have a history of ingesting large quantities of medication. Alternatively, BRASH syndrome patients are typically adherent to their prescribed medications.
Epidemiology
Underlying conditions & medications: This is usually seen in elderly patients with underlying cardiac disease being managed with a beta-blocker, diltiazem, or verapamil. Baseline renal dysfunction with lack of renal reserve function is often involved.
Trigger: BRASH syndrome is often initiated by hypovolemia (for example, due to gastroenteritis). Palmisano 2014 found that severe bradycardia was more common during the hottest summer months, a phenomenon they attributed to dehydration, hyperkalemia, and beta-blocker accumulation. Other triggers include up-titration of antihypertensives or potassium-sparing diuretics (e.g. spironolactone), any cause of hypoperfusion, or any cause of kidney injury.
Clinical presentation and diagnosis
There is a broad range of illness severity, extending from asymptomatic bradycardia to multisystem organ failure (e.g. shock, cardiogenic pulmonary edema, renal failure, shock liver). Patients often look better than their vital signs and laboratory studies would suggest. Regardless, bradycardia and/or hyperkalemia will typically cause alarm.
Treatment of BRASH syndrome
Some cases of mild BRASH syndrome may respond to basic medical therapies (e.g. IV calcium, fluid resuscitation). The discussion below is based on my experience in managing about a dozen of these cases in the ICU. As such, it is focused on more severe cases.
Hyperkalemia: Membrane stabilization & potassium shifting
The immediate management of hyperkalemia will involve IV calcium to stabilize the myocardium as well as IV insulin and dextrose to shift potassium into cells. Albuterol may be a good idea, both to shift potassium intracellularly and improve the bradycardia.
Fluid resuscitation if hypovolemic
Volume status varies greatly among different patients with BRASH syndrome. Some patients may be profoundly volume depleted, whereas others may present with volume overload due to anuric renal failure.
Fluid should be provided to patients with hypovolemia. Most patients with BRASH will have a combination of hyperkalemia and metabolic acidosis, making isotonic bicarbonate a good initial choice for fluid resuscitation. Once sufficient isotonic bicarbonate has been given to normalize the bicarbonate, additional resuscitation may be performed with lactated ringers. This strategy of pH-guided resuscitation is discussed further here.
Kaliuresis (+/- Dialysis)
Elimination of potassium usually involves potassium-wasting diuretics to facilitate potassium excretion in the urine. Patients who fail to produce urine in response to diuretics will require dialysis. Therefore, the initial attempt at diuresis is often very aggressive in an attempt to avoid dialysis:
In addition to diuretics, fludrocortisone may also facilitate renal potassium excretion. This may be especially useful in patients taking medications that reduce aldosterone levels (e.g. ACE inhibitors).
Catecholamine for bradycardia & shock
If the patient remains hemodynamically tenuous despite the above measures, there is often a temptation to wait and see if things will gradually improve. This is usually a mistake for three reasons:
- Diuretics work better if the kidney is being adequately perfused.
- Renal failure may resolve more rapidly if renal perfusion is established immediately.
- Bradycardic patients may have hypoperfusion out of proportion to their degree of hypotension. Occasional patients manage to compensate for severe bradycardia with vasoconstriction, thereby normalizing their blood pressure. However, their cardiac output is still low due to persistent bradycardia, based on the following equation: (5)
 For persistent instability, a catecholamine is usually helpful. There are a variety of choices for bradycardic shock, all of which seem to work (e.g. dopamine, epinephrine, and isoproterenol)(6). Some aspects of epinephrine and isoproterenol are notable:
For persistent instability, a catecholamine is usually helpful. There are a variety of choices for bradycardic shock, all of which seem to work (e.g. dopamine, epinephrine, and isoproterenol)(6). Some aspects of epinephrine and isoproterenol are notable:
Epinephrine: This may be a good choice for sicker BRASH patients. Low doses of epinephrine (e.g. 0-10 mcg/min) preferentially hit the beta-adrenergic receptors. Activity on the beta-1 receptors in the heart will increase chronotropy and isotropy. Meanwhile, the effect on beta-2 receptors tends to shift potassium into the cells, thus treating hyperkalemia.
Isoproterenol: This may be a reasonable option for less ill patients without central access. Isoproterenol is a nonselective beta-agonist, so it stimulates the heart and shifts potassium into cells (with less effect on potassium than epinephrine)(7). The advantage of isoproterenol is that it has no vasoconstrictive properties, so it is relatively safe to give via peripheral IV (8).
Treatments that usually aren’t necessary
Advanced therapy for beta-blocker or calcium channel intoxication: Usually patients with BRASH syndrome don’t have profoundly high levels of beta-blocker or calcium channel blocker. Therefore, they tend to respond to infusions of catecholamines as described above. I’ve managed only one patient who required high-dose insulin infusion and lipid emulsion therapy in this situation (he had paroxysmal AF and was receiving maximal doses of both beta-blocker and calcium-channel blockers)(9).
Glucagon: This isn’t an ideal treatment for these patients for two reasons. First, it tends to cause emesis and isn’t uniformly effective. Second, even if it does work, it is only a short-term solution. These patients typically require therapy for 12-24 hours, so infusion of catecholamines or milrinone is logistically more appealing (10).
Transvenous pacing: BRASH syndrome is really a metabolic/toxicological problem, which almost always improves with aggressive medical management. Emergent transvenous pacemaker placement is usually unnecessary (e.g. IV calcium is safer, faster, and more effective).
ACLS The ACLS bradycardia algorithm doesn't perform well in BRASH syndrome. Some patients fail to respond to atropine or transcutaneous pacing, but subsequently respond to IV calcium. Blindly following the ACLS algorithm may lead to over-utilization of transvenous pacing (11).
- BRASH syndrome refers to a vicious cycle which may occur when a patient taking AV node blockers develops renal failure and hyperkalemia. This leads to a spiral of worsening hyperkalemia, renal failure, and bradycardic shock.
- BRASH syndrome can be triggered by dehydration, medication up-titration, or any cause of hypoperfusion or renal dysfunction.
- The treatment of BRASH syndrome centers around aggressive therapy for hyperkalemia. Additionally, many patients require a catecholamine infusion to support their perfusion.
Related topics
- Hyperkalemia
- Management of severe hyperkalemia (PulmCrit)
- Treatment of severe hyperkalemia (EMCrit 2010)
- Fluid selection
- pH-guided resuscitation (PulmCrit)
- LR is safe in hyperkalemia (PulmCrit)
Notes
- My first case of BRASH syndrome was one of the more severe cases that I've encountered: an 80-year-old woman presented with pulmonary edema, renal failure, and shock requiring mechanical ventilation and high-dose vasopressors. The etiology of her multisystem organ failure was unclear, and it initially seemed like her case was hopeless. However, with aggressive supportive care she made a complete recovery. It was only in retrospect (after excluding all other possibilities) that beta-blocker accumulation was determined to be the diagnosis.
- Digoxin intoxication can occur via a nearly identical process, with a vicious spiral involving progressive renal failure and digoxin accumulation. However, the diagnosis and treatment of digoxin intoxication is rather different, so it isn't included here.
- References for this table are as follows: Aziz 2011, Erden 2010, Unterman 2008, Isabel 2006, Bonvini 2006, Vuckovic 2004, Zimmers 2002, Vazquez 1996, Jolly 1991, Lee 1986.
- There are reports of mild hyperkalemia causing bradycardia and heart block. I suspect that these patients may have underlying conduction system disease, but this remains an assumption.
- These patients are basically behaving similar to a septic shock patient treated with excessive doses of phenylephrine (a pure vasoconstrictor). The vasoconstriction improves the blood pressure, but it may actually worsen systemic perfusion (by increasing afterload on the heart). Thus, excess vasoconstriction creates a situation where the blood pressure looks OK, but the patient may be substantially malperfused. Clinical signs of this physiology at the bedside include cold extremities and poor urine output.
- Note that bradycardia may respond only to a combination of catecholamines and calcium. If bradycardia is refractory to catecholamines, consider giving additional calcium.
- At equivalent levels of beta-1 stimulation, epinephrine appears to have a greater effect on beta-2 receptors and potassium levels (Brown 1983). Although isoproterenol can cause hypokalemia, this magnitude of this effect may not be clinically significant at doses used for treatment of moderate bradycardia.
- There are no published case reports of skin necrosis from isoproterenol extravasation in PubMed. Given that isoproterenol causes vasodilation, it has actually been tested in animal models an antidote to treat extravasation reactions. Several references state that isoproterenol can be given peripherally (e.g. the Alfred ICU, Critical Care Study Guide by Criner GJ et al., and Critical Heart Disease in Infants and Children by Nichols DG et al.). The drawback from having no vasoconstrictive properties is that theoretically isoproterenol could decrease the blood pressure (due to peripheral vasodilation). Overall, the chronotropic and inotropic effects of isoproterenol seem to outweigh its vasodilatory effects, leading to an overall stable or improved blood pressure. Nonetheless, epinephrine may be superior to isoproterenol for patients with profound hypotension. When in doubt, isoproterenol can be started cautiously with additional medications on hand (e.g. epinephrine) to salvage the patient in case there is hypotension.
- This patient went from being profoundly ill with refractory shock to being asymptomatic and entirely stabilized within 18 hours. The only invasive procedures required to treat him were a central line and arterial line, placed emergently in the groin and then removed within 24 hours (technique described here). This is why BRASH syndrome is satisfying to treat: patients may arrive profoundly ill, but improve rapidly with aggressive medical therapy.
- It is possible to run a glucagon drip, but pharmacists have never been too keen on this idea (e.g. it would exhaust the hospital's glucagon supply). Glucagon bypasses the beta-receptor to increase myocardial cAMP levels. Milrinone has similar effects, and might be a logistically superior approach to accomplish this.
- Does anyone understand why IV calcium isn't in the bradycardia algorithm? The only reason I can think of is that they were worried about “stone heart” phenomenon if calcium were given to a patient with digoxin toxicity (a phenomenon of questionable validity). IV calcium and intralipid might be reasonable 3rd and 4th line medical therapies, after failure of atropine and catecholamines.
Opening image: https://commons.wikimedia.org/wiki/File:Occluded_mesocyclone_tornado2_-_NOAA.jpg
- Pulmcrit wee: The cutoff razor - April 15, 2024
- PulmCrit Blogitorial – Use of ECGs for management of (sub)massive PE - March 24, 2024
- PulmCrit Wee: Propofol induced eyelid opening apraxia – the struggle is real - March 20, 2024





I have treated two BRASH presentations this year in the field. Had remembered reading about this several years ago. The first case we followed ACLS and arrived in the ED with no real improvement. After following up with the Attending I realized what we had done wrong The second case we started Calcium, dextrose and Albuterol in transport to the ED. We were obtaining slightly increased pressures at arrival. I have been following EMCrit for about 3 years now and have been recruiting my fellow paramedics to the show. I appreciate the detailed information presented. It helps me be a… Read more »
Fantastic, great stuff. Once you become aware of this entity, you start recognizing it much more frequently.
My question is trimmed. The larger and smaller signs are the culprit of the problem
It should be:
Do you mean you still give IV calcium even if K less than 6.0?
How does this work? as to my understanding, calcium acts to stable the cardiac membranes.
And we only usually give calcium when K more than 6.0 in our locality.
Great article! This is the first time I’ve come across the mnemonic though I’ve had multiple patients with profound bradycardia in renal failure with profound acidemia and hyperkalemia (though I honestly don’t remember if they were on av-nodal blockers at the time). What I found usually works is bicarb, and lots of it; I titrate until I see an improvement in the rhythm like appearance of p waves or qrs narrowing if wide initially–usually this is 5-6 amps immediately once iv access has been established, then start on bicarb drip and titrate to a pH of 7.3–this significantly improves the… Read more »
I am wondering at what juncture should we consider giving IV calcium…
Good question. I’m not aware of any great evidence on this (would be very difficult to study). My practice is to give IV calcium if there are EKG changes or if the potassium is very high (perhaps >7, but this is somewhat arbitrary). In some cases, calcium will be given empirically as a diagnostic/therapeutic attempt to treat bradycardia due to *possible* hyperkalemia before the labs are back. Patients with severe hyperkalemia may require lots of calcium. So, if you’re treating a bradycardic patient and the first gram doesn’t work, they may simply need more calcium. Fortunately hypercalcemia seems to be… Read more »
Thanks for the reply Josh!
I see that you have mentioned: Patients with BRASH syndrome may have only mild hyperkalemia, with bradycardia due to synergy between hyperkalemia and the AV node blocker
What do you mean by mild hyperkalemia in your clinical setting? Mine’s K ~5.5 and is regarded to be mild already.
So are you saying that even earlier IV Calcium is warranted when K <6 in treating BRASH patients?
I am bit confused here.
Awesome post! I am also curious as to why calcium was removed from the PEA algorithm.
I believe there was some anecdotal evidence that calcium chloride attenuates the beta-adrenergic properties of epinephrine. Any further info on this?
Is there any place for dobutamine in the treatment of BRASH?
Very happy to see you reiterate that an epinephrine infusion at <10 mcg/min is a great inotrope.Once you get higher than 10 mcg/min you start hitting more alpha-adrenergic receptors and see more acidosis and gluconeognesis. I think epi infusions get a bad rap mostly due to improper use.
Thanks. With regards to the calcium/epinephrine interaction: there was a report that calcium inhibited some effects of epinephrine in 1990 (PMID 2153475) but this wasn’t validated by a subsequent study (PMID 1734794). I’m not aware of this being a big problem. I have used dobutamine in BRASH. This may be a good approach to treating a patient with poor perfusion from bradycardia, especially if their blood pressure is on the normal or higher side (noting that dobutamine may cause some vasodilation which could tend to decrease the blood pressure a bit compared to agents like dopamine or epinephrine). Yep, I… Read more »
Yes, I would certainly agree that epinephrine has better inotropic properties and less arrhythmogenicity than similar doses of dobutamine. I believe dobutamine has it’s place in therapy but with escalating doses it quickly becomes less useful. Overcoming the dogma that epi infusions will cause all sorts of bad juju is sometimes difficult. Also titrating epinephrine to cardiac output rather than BP can be difficult to educate on. A combination of milrinone and low-dose epinephrine infusions may be useful in situations of beta-receptor downregulaton or beta-receptor saturation. This combo is also useful in RV failure. Thanks for the reply and interesting… Read more »
Vascular Care Center at Somajiguda, Hyderabad is a specialized hospital for treatment for vascular and endovascular diseases. These type of diseases are occurred due to the irregularity of the circulatory system, arteries and veins create a problem in blood flow direction. If the disease is in serious stage surgery treatment is required. Doctors at Vascular Care Center are efficient and skilled surgeons. Dr.G.V. Praveen Kumar is one of the eminent surgeons at Vascular Care Center who performed many thousands of vascular system related surgeries. A Vascular (AV)access are of two types for permanent access Arterio Venous (AV) Graft and Arterio… Read more »
dfdshfgjhrhe
My father just died from this, or at least that’s what i was told. He entered the hospital and an hour later he was gone. Can anyone please explain to me how this could happen?
Had a patient present to ED after feeling well day prior- came in obtunded, K+ 7.1, kidney function poor. Afib slow response 30s; BP 60s-70s/palp. Takes dig, cardizem, metoprolol- seems like they definitely fit the bill! My first experience with a BRASH-type patient and never knew of this until reading your blog post, very interesting. Looks like they turned around the next day! Thanks for all you do! FOAMed has taught me so much
Who came up with the acronym of BRASH and when ?
Thank you Josh for this concept. I think the main point here is that one should impact on every component of vicious circle, not the only one most striking lab sign. In fact it’s applicable more generally to clinical medicine. But I want to make some specific comment on this: “Transvenous pacing: BRASH syndrome is really a metabolic/toxicological problem, which almost always improves with aggressive medical management. Emergent transvenous pacemaker placement is usually unnecessary (e.g. IV calcium is safer, faster, and more effective).” I wouldn’t completely agree. Bradycardia is major component of BRASH-syndrome and its quick and safe (most safe indeed, sorry,… Read more »
[…] Farkas J. BRASH syndrome: Bradycardia, Renal failure, Av blocker, Shock, and Hyperkalemia. Pulmcrit.… […]