
Happy hypoxemia is severe hypoxemia (poorly responsive to supplemental oxygen) without dyspnea. This isn’t anything especially new – we have occasionally seen this since time immemorial. However, COVID is causing us to re-think how to manage this physiology.
understanding the paradox of happy hypoxemia
The key to understanding this is thinking about oxygenation and CO2 clearance separately. These problems typically run together, but under certain situations they can become divorced.
(1) oxygenation
Hypoxemia occurs when blood somehow passes from the right ventricle into the left ventricle without being fully oxygenated. This generally occurs in one of two ways:
- Ventilation-perfusion mismatch (V-Q mismatch): Some parts of the lung have excess ventilation, whereas other parts have inadequate ventilation. Blood going to portions of the lung which are inadequately ventilated will be starved of oxygen. Alternatively, blood flowing to areas of the lung with excess ventilation will not be saturated over 100%. Taken together, the net result is hypoxemia. Increasing the concentration of inhaled oxygen generally fixes this problem – because even poorly ventilated areas of the lung will now get enough oxygen.
- Shunt physiology: Blood flows from the right ventricle to the left ventricle without ever coming into contact with oxygenated alveoli at all. Examples include an anatomic abnormality (e.g., ventricular septal defect) or complete dysfunction of parts of the lung (e.g., mucus plugging of one lobe of the lung). A hallmark of shunt physiology is that it is poorly responsive to increased levels of oxygen (because blood isn’t coming anywhere close to ventilated alveoli). Thus, any patient who is desaturating despite high concentrations of inhaled oxygen likely has a shunt.
(2) CO2 clearance & dead space
CO2 clearance depends on the amount of gas entering and leaving the lungs every minute, which removes CO2 from the body.
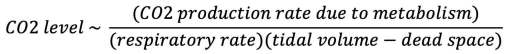
A key variable here is dead space – which is gas that is inhaled and exhaled but which does not participate in CO2 clearance. Dead space is essentially wasted breath – the body moves gas in and out of the chest, but it achieves no CO2 clearance. Excess dead space can arise at a “micro” level or a “macro” level:
- “Micro” level dead space – scarred alveoli due to ARDS become inefficient at CO2 clearance. For any given tidal volume, there is less effective CO2 clearance.
- “Macro” level dead space – imagine that a patient suddenly develops a segmental pulmonary embolism. Gas still goes in and out of that lung tissue, but no CO2 clearance occurs.
(3) work of breathing, dead space, lung compliance & airway resistance
The work of breathing is most strongly affected by the drive to clear CO2. This is influenced by:
- Dead space – increasing the dead space means that the patient must inhale and exhale more gas every minute to maintain the same CO2 level.
- Lung compliance – if the lungs have more elastic recoil impeding inflation, this will increase the work of breathing.
- Airway resistance – any obstruction to airflow, such as asthma, may increase the work of breathing.
(4) how happy hypoxemia occurs
Refractory hypoxemia with a normal work of breathing can occur if all of the following conditions are met:
- A right-to-left shunt is present.
- There isn’t excessive dead space.
- Compliance and resistance of the lungs are reasonably normal.
The most simple example of happy hypoxemia is a patient with an intra-cardiac right-to-left shunt. Such a patient may have normal lungs and thus no difficulty clearing CO2 (with a normal work of breathing). However, the patient may be quite hypoxemic. This represents perhaps the most pure divorce between oxygenation and CO2 clearance.
Now let’s take things a step further. Imagine two patients, with X-rays as shown below. One patient had a left lower lobe resection, whereas the other patient has complete left lower lobe atelectasis (causing the left lower lobe to collapse into a tiny nubbin hiding behind the heart). In both of these patients, the remaining lung tissue is normal.

Neither of these patients has extra dead space – in both cases, the lung tissue receiving ventilation is normal. Both patients have sufficient lung tissue remaining so that their overall chest compliance is close to normal. Thus, both patients can clear CO2 without difficulty – so they won’t be dyspneic.
However, the patient with left lower lobe collapse will shunt deoxygenated blood through the collapsed lung tissue – leading to hypoxemia. Thus, complete atelectasis can cause a happy hypoxemia phenotype – isolated hypoxemia with a normal work of breathing.
An additional quirk of physiology bears mention here – the ability of hypoxemic vasoconstriction to compensate for areas of the lung which are dysfunctional. For example, if the patient with atelectasis is able to strongly vasoconstrict blood flow to the collapsed lung, this may prevent significant shunting. Different patients seem to have varying abilities to compensate via hypoxemic vasoconstriction (e.g. patients with cirrhosis are often poor at hypoxemic vasoconstriction).
Recent case at Genius General Hospital illustrating happy hypoxemia
A woman presented to Genius General with a chief complaint of a wrist laceration. Further history revealed that she had been falling down repeatedly at home. Vital signs showed that she had a room air saturation of 70%! Despite this, she had absolutely no respiratory complaints.
She had a fever, leukocytosis, lymphopenia, and B-lines bilaterally. Thus, she was diagnosed initially with probable COVID-19. She remained hypoxemic despite 100% high-flow nasal cannula oxygen, leading to ICU admission.
This is her X-ray:

This doesn’t look like a COVID chest x-ray at all. Based on this X-ray, a repeat focused chest ultrasound was performed, which revealed a densely consolidated right lower lobe with dynamic air bronchograms – suggestive of a lobar bacterial pneumonia (she had already been started on empiric ceftriaxone and azithromycin, so her antibiotic coverage was quite adequate).
(Incidentally, this reveals the strength of chest X-ray during a COVID epidemic. Yes, lung ultrasound is very good. But a chest X-ray allows you to check your work. Especially if you’re moving quickly and blinded by availability bias, a chest X-ray will help prevent you from missing things. So, to all the ultrasonography purists out there – I’m sorry, but I still like admission chest films.)
So, even in the middle of a COVID pandemic, this patient presented with a happy hypoxemic presentation due to lobar pneumonia! She was shunting blood through the consolidated lung tissue, causing hypoxemia. Her remaining lung tissue was essentially normal, allowing her to clear CO2 without much difficulty (hence the lack of dyspnea). This is essentially the same as the patient with complete lobar collapse discussed above.
Based on our evaluation, we were increasingly suspicious that she had a lobar bacterial pneumonia (rather than COVID). The next question arose – how to manage her hypoxemia?
Before the COVID pandemic, I would have intubated this patient due to bacterial pneumonia with refractory hypoxemia. Without thinking twice. That was the textbook management of this condition. In 2018, there would have been 100% consensus that this patient required intubation.
But… this is April 2020. And she looked really good. Her saturation was dipping into the low 80s on 100% high-flow nasal cannula, but she was quite comfortable. We decided to try awake proning, and this caused her saturations to rise into the 90s. She continued to be comfortable. We weaned back the FiO2 to 90%.
Over the next day, her saturation continued to rise and fall, at times dipping into the low 80s. Her overall trajectory was one of recovery, however. She gradually improved and didn't require intubation.
Eventually her labs showed pneumococcal bacteremia. Her COVID PCR came back negative. So she did indeed have a classic, timeless presentation – lobar pneumococcal pneumonia. Treated with awake proning and permissive hypoxemia. What’s old is new again. (I have used awake proning for occasional patients with viral pneumonia or interstitial lung disease since 2016, but not usually for lobar pneumonia).
Approach to hypoxemia: avoiding a paint-by-the-numbers strategy
Whenever possible in medicine, triage and treatment decisions should be based upon an understanding of the physiology and disease process (rather than isolated numbers). For example:
- Saturation of 85% in a patient with asthma on high levels of FiO2 suggests severe hypercapnia or mucus plugging – which is extremely concerning and may predict deterioration.
- Saturation of 85% in a patient with a lobar shunt due to pneumococcal pneumonia who is on appropriate antibiotics is less worrisome – the natural history of this disease is often gradual resolution. As long as the patient is comfortable and defending their CO2, intubation is less likely to be required.
- Saturation of 85% may be a chronic feature in a patient with advanced congenital heart disease or interstitial lung disease.
Thus, the saturation number (85%) means little in isolation. Clinical context is required to understand what this means. Approaches to intubation or patient triage based solely on saturation or ABG values are doomed to failure.
What is the pathophysiology of happy hypoxemia in COVID-19?
So, the physiology of happy hypoxemia in COVID-19 likely involves the following elements:
- Intra-pulmonary shunting
- Relatively preserved lung compliance
- Lack of excessive dead space
- (Possibly also: dysfunctional hypoxemic vasoconstriction)
This is consistent with experience in mechanical ventilation of intubated patients with early COVID-19. Specifically, the lung compliance seems to be relatively normal. Additionally, there doesn't seem to be increased dead space (so CO2 clearance isn't a problem).
The precise mechanism behind this physiology is currently unknown (because autopsy studies typically involve patients who have been intubated for a while, leading to superimposed ventilator-induced lung injury). My best guess is that the pathophysiologic mechanism might involve:
- Most of the lung tissue functions normally.
- Some alveoli receive no aeration, causing them to function as a shunt. My guess is that this could result largely due to atelectasis (given that early APRV, proning, or CPAP seem to improve oxygenation substantially).
- Lack of substantial pulmonary embolism or microvascular thrombosis (these can occur, but seem to be a delayed feature of COVID-19).
This is generally compatible with the concept of malignant atelectasis explored earlier:

- Happy hypoxemia (severe hypoxemia without dyspnea) can be generated by a combination of shunt physiology, preserved lung compliance, and lack of dead space. This may result from any lung disease which causes a limited amount of shunt, while preserving the remainder of the lung (e.g., lobar consolidation or atelectasis).
- Happy hypoxemia has existed forever, but these patients presented only occasionally. COVID has forced us to re-think our approach to treating this physiology.
- When considering a patient with hypoxemia, the underlying pathology and physiology of hypoxemia is often more important than the exact saturation. Clinical context predicts the likelihood of deterioration or improvement.
- Reconceptualizing oxygen saturation will change the way we practice critical care – even long after the epidemic has passed.
- Pulmcrit wee: The cutoff razor - April 15, 2024
- PulmCrit Blogitorial – Use of ECGs for management of (sub)massive PE - March 24, 2024
- PulmCrit Wee: Propofol induced eyelid opening apraxia – the struggle is real - March 20, 2024
Thanks for all your contributions to FOAM. What are your thoughts on etiology of malignant atelectasis in covid-19? Loss of surfactant from viral effect on pneumocytes?
Seems like it would make sense. I haven’t read anything where surfactant has been a modality studied yet. Dr Farkas, any insight on surfactant being beneficial in these patients?
Excellent!! good to discuss this !! The concept that is proposed is very interesting. I have some doubts / questions, but it is very good. It reminds me of cirrhotic patients with pulmonary arterio-venous fistulas. These patients always have hypoxemia! They do not have hypercapnea, and they do not have dyspnea, they have “normal” lungs, without collapse. But I do not agree with some things that the article proposes. The characteristics of the phenomenon that explains the hypoxemia generated by a purely vascular R-L intra-pulmonary shunt are different from those of hypoxemia generated by alveolar occupation. In the first, there… Read more »
Good to see that the concept of understanding “the underlying pathology and physiology ” is back . I just hope that we will re-think our “protocols” of just chasing “numbers” and much of what we all do.
“Benign negligence” many times is better.
Thank you! Been teaching this for years to get EMS, primary and secondary care staff to spot pneumonia in children, when the absence of recession/ retractions fools them into thinking the kid is ok. Measure RR and SaO2 and you’ll spot that child!
Thank you for sharing this with us, but I have couple of comments; First, one would expect hypoxemia to be a driving factor for increased respiratory drive even in the context of normocapnea, so, how can we explain the absence of this especially in the context of acute setting to which the body has no time to acclimatize such like in chronic hypoxemic state ? Second, compliance in this cluster of patients in fact is not entirely normal, from my observations, it seems to be mildly to moderately reduced, and obviously this depends on the stage of the disease, but… Read more »
COVID patients are suffering from hypersensitivity pneumonitis due to histamine released by
neutrophils in the lungs. HP has been associated with thrombic events in a number of studies..
Said HP will continue until the patient has cleared the majority of the viral load from the lung tissue; the greater the viral load, the greater the symptoms of HP.
The patient needs to be treated with H1 and H2 antagonists to reduce EVLW and ibuprofen to reduce pulmonary hypertension – which will also reduce EVLW and improve hypoxia.
Take a look at the acute presentation of hypersensitivity pneumonitis; it’s a COVID patient. Hypoxia at rest, ground glass CT, fever, maliase, cough, shortness of breath.
Then look at recent research that shows that neutrophils produce histamine in response to pathogens in the lungs: (something we didn’t even know until a decade ago)
https://www.ncbi.nlm.nih.gov/pubmed/23572231
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3205215/
The patients need to be treated with antihistamines and anti-inflammatory medication until their body can clear the remaining viral load from the lungs.
As a resident I have these same questions. Would be delighted to have some answers to that.
Thank you
Suppose the “Happy Hypoxemic” patient’s hypoxemia is secondary to shunting from lobar consolidation / atelectasis as well as increased circulating vasodilators / decreased clearance of circulating vasodilators (as in hepatopulmonary syndrome), why not try treating these patients with Methylene Blue?
Sounds like it’s time for the adult cc docs to take a lesson from us on the peds side of things. We’ve been playing with happy blue babies since forever. The key questions one has to ask themselves: 1. How does the patient look? Is she anxious/air hungry/distressed? Or is she cooing at herself in the mirror and giggling when you make funny faces? 2. Is there any evidence of end organ dysfunction or concerns for inadequate O2 carrying capacity? Although it’s not clear what the lowest tolerable Hb in a patient with cyanotic congenital heart disease is, most of… Read more »
Great post, as always. I’d like to know your opinion regarding some points: – Pulmonary embolism and dead space: yeap, segmental PE stops perfusion and it increases dead space, but often the main problem is hypoxemia rather than hypercapnia since you have a V/Q mismatch due to increased blood flow through healthy lung (if Q remains the same, you have the same amount of blood passing across a smaller vascular pulmonary bed so in this sense you have a V/Q mismatch) http://www.ventilab.org/wp-content/uploads/2017/01/embolia-300×295.jpg . – A more common reason for increased dead space in ICU is hypotension associated with higher ventilatory… Read more »
Thanks for the great article! I’m still a little unclear about how you can have adequate clearance of pCO2 with simultaneous inadequate absorption of O2. In the example of the intra cardiac right-left shunt would you not also get accumulation of CO2 which would lead to dyspnea? I’m almost certainly missing something….
CO2 diffuses across the capillaries about 20x faster than O2 due to its solubility
You are simply misinterpreting readings from pulse oximeters. Oximeters do not take into account RBC count, or hematocrit levels or Hgb. Hypoxemia is not the same as hypoxia. With larger hematocrit/RBM levels your patients can cope with both CO2 and O2 exchange.
An acclimated climber on mount Everest can show PaO2 = 52% but he is not hypoxic.
We are rapidly transitioning to high flow nasal cannula and tolerance of hypoxia on the relatively well appearing patients. As we do this we have a limited number of NFNC and are still trying to avoid CPAP/ BIPAP given limited negative airflow rooms. Is there any reason why the use of the CPAP -> viral filter -> BVM – > high O2 set up can’t be left in place for days on end? This set up has been described as a pre-oxygenation strategy before intubation steps but seems if someone turns around on this maybe this can be continued for… Read more »
Your patients are not literally “hypoxic”, they just have lower PaO2 saturation, BUT they have elevated hematocrit, RBC counts, and hemoglobin. They can tolerate low saturation levels because RBM (red blood mass) is substantially higher than is normal for them.
Pulmonary physiology 1. Air hunger is caused by acute CO2 retention / acidosis but not by hypoxia. Anyone who has been at altitude and had a sat of 80% knows this. Rapid ascent to 8000m can kill you but you will not feel bad until you black out. Excellent post -thanks.
When PaO2 falls below 60 mm Hg (<90% sat) hypoxia becomes a progressively significant stimulus to ventilation (via peripheral chemoreceptor mechanisms). I'm wondering why this would not be the case in the COVID-19 lung., which the patient would then sense as dyspnea. Maybe it is not perceived as dyspnea by the patient because lung compliance is preserved, and thus WOB feels normal? Is PaCO2 typically low in the happy hypoxemia scenario? This would be consistent with a hypoxic stimulus to ventilation. Additionally, hyperventilation would produce an alkalemia, which would lessen ventilatory drive-at least in the short term.
Pulse oximeters don’t take into account increased RBM, because they assume normal Hgb, and normal RBC counts. These patients had been hypoxic for a while, before seeking medical help, and have compensated/acclimated via erythropoiesis.
SaO2 < 90% are therefore misleading.
Pulse oximetry is unreliable in patients with high hematocrit. Notably, the papers cited below indicate that pulse oximetry’s accuracy improves with lower hematocrit percentages.
The Accuracy of Pulse Oximetry at Two Haematocrit Levels
M Vegfors 1, L G Lindberg, P A Oberg, C Lennmarken
https://pubmed.ncbi.nlm.nih.gov/1632169/
Accuracy of Pulse Oximetry at Various Haematocrits and During Haemolysis in an in Vitro Model
M Vegfors 1, L G Lindberg, P A Oberg, C Lennmarken
https://pubmed.ncbi.nlm.nih.gov/8331993/
When PaO2 falls below 60 mm Hg (<90% sat) hypoxemia becomes a progressively greater stimulus to ventilation (via peripheral chemoreceptor mechanisms) which would be reflected by a low PaCO2. Are early stage COVID patients often hypocapneic? It seems patients with severe hypoxemia would sense an increased ventilatory drive. Perhaps it is not sensed as an increased WOB and dyspnea because lung compliance is near norma?. Additionally, chronic hypocapnea would produce an alkalemia, which would suppress the hypoxic ventilatory stimulus Great post!.
I was exactly thinking so..I could even not find sufficient data and articles related to blood gas and pH parameter in such cases. Hypocapnia and related pH change for happy hypoxia needs to explore or if there’s the answers, it will be extremely helpful if shared.
You can test for “silent/happy hypoxemea” (hypoxia is different from hypoxemia) by blood tests used for polycythemia vera, 1) elevated RBC counts (and possibly platelets) 2) elevated Hgb, 3) elevated hematocrit
4) as opposed to polycythemia, however, erythropoietin levels should be higher, NOT LOWER.
Assuming that covid pneunomia is resolved already, higher than normal hematocrit/RBC counts can be treated with a simple blood donation. This would prevent unavoidable extensive hemolysis which would lead to massive vasculitis and damage.
With “happy” hypoxemic patients, in the “oxygen first” approach discussed in recent Emcrit webinar, are we not concerned with oxygen toxicity? If so, what’s your thoughts on why that may be the case? Or if not, is the benefits out weigh the risk here? How long would you leave someone on 100% FiO2 (or >60), whether they are on high flow or on the vent with these L-type high compliance patients?
Is it possible that the pulmonary shunting is due to localized inflammation caused by histamine production? I’ve been working on a theoretical treatment that may be relatively simple to try or expand on. To summarize, the theory proposes that the cause of acute lung injury is due to histamine release within infected lung tissue and focuses on attenuating symptoms related to that histamine release. This treatment does nothing to ameliorate viral load, but rather focuses on addressing the subsequent ARDS/sepsis/multi-organ failure that some patients progress to, so it could be administered concurrently with anti-viral treatments. Please also note that data directly… Read more »
In this patient with a right lower lobe pneumonia, would turning to the left lateral position also result in improved oxygenation? Perfusion redistributes to the normal lung, with better V/Q matching. It may also be easier for the patient to lie on the side compared to lying prone for extended periods of time. Could the vasoplegic state and the abolition of hypoxic pulmonary vasoconstriction be due to the virus occupying the ACE II receptors, leading to attenuation of the effect of angiotensin II? Or could it be vasodilatation that results from the intense cytokine storm that is often seen in… Read more »
in response to your comment-“The precise mechanism behind this physiology is currently unknown (because autopsy studies typically involve patients who have been intubated for a while, leading to superimposed ventilator-induced lung injury).” please note these biopsy findings published by incidental covid infection seen from routine biopsy for lung cancer patients early in their covid infection https://www.jto.org/article/S1556-0864(20)30132-5/fulltext?mobileUi=0 Figure 3 Histologic changes of coronavirus disease 2019 pneumonia in case 2. (A) Evident proteinaceous and fibrin exudate; (B) diffuse expansion of alveolar walls and septa owing to fibroblastic proliferations and type II pneumocyte hyperplasia, consistent with early diffuse alveolar damage pattern; (C) plugs of… Read more »
Hey Dr Farkas, I was wondering about what was mentioned about the unanimous decision to intubate with refractory happy hypoxemia in the pre-COVID era. Im curious, since in my few short years of attending time over the last 3 years, I have had success in staving off intubations in comfortable patients by getting them into an ICU setting while on BiPAP, despite it being a more long term pathology like pneumonia. I found myself preoxygenating them with NIPPV with the possibility of intubating them, but then would find them tolerating well, and comfortably, and allowing them to continue that course… Read more »
The author has not talked about O2 carriage. This again is one of the theories about hypoxia in Covid, the virus displacing O2 from it’s binding sites in Hb. This area needs to be looked into as well
Arun I wondered about this as well has any medic done research on this ????
What effect has covid 19 virus have on the capillaries surrounding the alveoli and is there any adverse effect on the haemaglobin oxygen capacity of the red blood cells in this DISEASE In Covid 19 Patiemts are the ferritin levels higher than normal and if so why?
Hypothetically, if we could introduce oxygen into the bloodstream without using the lungs or heart, do you think that would have a positive effect on the co-morbid systemic pathology that occurs with happy hypoxemic patients?
Thanks for the possible hypothesis. I work Clinically with COVID patients – who are recovering and getting rehabilitated – they show this happy hypoxia with exertion or activities, reversed by supplementary oxygen.
Noticed this phenomenon slowly resolving with regular functional rehabilitation with Successful weaning of supplementary oxygen in 2-3 weeks.
Oxygen saturation reading for a healthy person (already acclimated) in Denver is 90% It is not because his lungs are damaged, it is because RBM (red blood mass) is higher than at sea level. On mount Everest SaO2 reading was 54% in an acclimated climber. Your conjectures about the mechanism are plausible, but using Occam’s razor increased RBC mass is far simpler and thus likely the correct physiology. Modern oximeters assume normal Hgb levels and provide data that is misleading although paradoxically accurate. The reason you see “happy hypoxemia/hypoxia” is because enough oxygen is being delivered with partial RBM. Testing… Read more »
Mechanisms of hypoxemia Malay Sarkar, N Niranjan, and PK Banyal https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5234199/ Hypoxic regulation of erythropoiesis and iron metabolism Volker H. Haase https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2904169/ Hypoxia Signaling Cascade for Erythropoietin Production in Hepatocytes Yutaka Tojo, Hiroki Sekine, Ikuo Hirano, Xiaoqing Pan, Tomokazu Souma, Tadayuki Tsujita, Shin-ichi Kawaguchi, Norihiko Takeda, Kotaro Takeda, Guo-Hua Fong, Takashi Dan, Masakazu Ichinose, Toshio Miyata, Masayuki Yamamoto, Norio Suzuki https://mcb.asm.org/content/35/15/2658 Hypoxia Induces a Prothrombotic State Independently of the Physical Activity Marisa Ninivaggi,Marieke de Laat,Marcus M. D. Lancé,Cécile H. Kicken,Leonie Pelkmans,Saartje Bloemen,Marlou L. Dirks,Luc J. C. van Loon,José W. P. Govers-Riemslag,Theo Lindhout,Joke Konings,Bas de Laat https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0141797 Hemolytic vascular inflammation: an… Read more »
Hey. First at all, Thanks for ur contributions. This may explain why some COVID19 patients have good response to higger peep. Have a question. What’s your opinion about the use of PD3 inhibitos, nitric oxide even penthoxiphiline as adyuvants for this guys with prolongued AaDO2 and suspicious of microtrombi formation in pulmonary capillaries.?
I have found that the COVID pts admitted to the medsurg unit have this condition. I’ve never heard the term “happy hypoxia” but I have been looking for information on “silent hypoxia”. The patients that are being admitted for COVID are presenting with hypoxia but then weaned to room air because their spot pulse ox shows a saturation above 90%. Still marginal but seemingly adequate to wean from supplemental O2. The problem is that these patients don’t necessarily complain of SOB but rather lethargy and weakness. When you check their O2 sats they have desat to below 80…silently. We have… Read more »
I suffered deeply with stage four sarcoidosis which progressed to idiopathic pulmonary fibrosis (IPF). I was hospitalized eighteen times in three years, mostly with flare ups and pneumonia. For me, the chronic widespread pain takes my breath away more than the disease itself. At times the pain is so severe I find it difficult to do anything. My life was gradually coming to an end, until I saw World Herbs Clinic on google during my research for alternative help for IPF.. I knew health is wealth thus I decided to give it a try, thank God I did. I got the… Read more »
Thanks for making this easy to understand.
absolutely great article, answering all of my questions