
Over the last several weeks, we have seen a flurry of studies on various anti-viral therapies (lopinavir-ritonavir, hydroxychloroquine, chloroquine, and remdesivir). On the whole, these studies were consistently unimpressive. Perhaps remdesivir works a bit, but overall these studies simplified management by allowing us to ignore several drugs.
I’m expecting the anti-inflammatory trials to be the opposite of this. COVID-19 is a highly inflammatory disease process, so it’s possible that a variety of different anti-inflammatory agents will exhibit some efficacy against it. So, we may now see a spate of positive trials involving various agents. This is good, but it may also lead to enormous confusion. If anakinra and tocilizumab both show efficacy, should we use one? Or both? Who should receive them? How should we manage limited supplies and high costs of these medications?
Cavalli G et al: Interleukin-1 blockade with high-dose anakinra in patients with COVID-19, acute respiratory distress syndrome, and hyperinflammation: a retrospective cohort study
This is a single-center before/after study design evaluating the use of high-dose anakinra for patients with COVID-19 and hyperinflammation.1 Anakinra is an IL-1 receptor antagonist which is a front-line therapy for hemophagocytic lymphohistiocytosis (HLH). To the extent that COVID-19 resembles HLH, anakinra might be expected to be efficacious. IL-1 levels are elevated in COVID-19, along with many other pro-inflammatory cytokines. Anakinra is generally regarded as a fairly safe agent, but it is expensive and in limited supply. The use of anakinra for sepsis/HLH overlap syndrome has been previously discussed on the blog here (it showed promise in a retrospective re-analysis of a Phase-III clinical trial).2
Important inclusion criteria were as follows (these criteria were used prospectively to determine candidates for anakinra, and later applied in a retrospective fashion to select patients in the control group).
- Patients on noninvasive ventilation (intubated patients were excluded).
- Moderate-to-severe ARDS (P/F ratio < 200 on noninvasive ventilation with at least 5 cm PEEP).
- Hyperinflammation (defined as C-reactive protein at least 100 mg/L, ferritin at least 900 ng/ml, or both).
- No evidence of bacterial infection.
- No concomitant use of other anti-inflammatory agents or steroid.
- No enrollment in another clinical trial.
Three cohorts of patients exist:
- “Standard treatment group” – 16 patients who received no anakinra between 3/10-3/17.
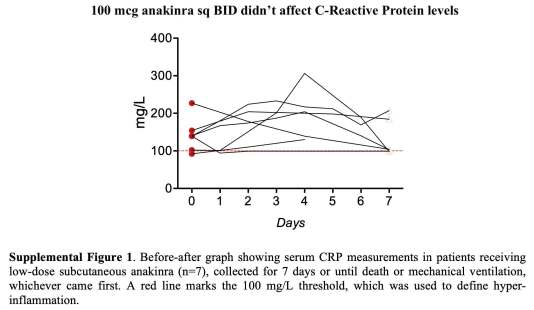
- Low dose anakinra – 7 patients who received 100 mcg anakinra subcutaneously BID between 3/10-3/17.
- High dose anakinra – 29 patients who received 5 mg/kg anakinra intravenously BID between 3/17-3/27. Treatment was continued until there was sustained clinical benefit (defined as a 75% reduction in serum C-reactive protein and sustained respiratory improvement with P/F > 200 for at least two days), death, bacteremia, or side-effects (e.g. transaminases >3-fold the upper limit of normal). Following discontinuation of high-dose anakinra in patients who had improved, 100 mg anakinra subcutaneously BID was continued for three days to prevent rebound of inflammation.
The low dose anakinra didn’t appear to work (based on a lack of effect on C-reactive protein levels; figure below). Thus, this therapy was abandoned in favor of higher doses provided intravenously. The study focuses on comparison of standard treatment versus high-dose anakinra.
The standard treatment and high-dose anakinra groups appear reasonably well matched at baseline:

Patients treated with high-dose anakinra had better survival rates and likelihood of respiratory improvement:

Use of high-dose anakinra appeared to cause more consistent reduction in C-reactive protein levels and improvement in oxygenation:

Anakinra appeared to be generally well tolerated. It was discontinued in four patients due to bacteremia and the concern that anakinra might suppress the response to infection. In three patients, anakinra was discontinued due to increases in transaminases greater than three times the upper limit normal. No signs of rebound inflammation were noted. Similar rates of infection and transaminitis were observed in patients receiving standard treatment, suggesting that anakinra didn’t cause these abnormalities.
Limitations of the study
- As a retrospective before/after study, this is subject to multiple sources of bias which prevent it from proving causality. For example, it’s possible that subtle improvements in the care of COVID-19 patients caused more recent cohorts of patients to fare better.
- During 3/10-3/17, some patients were treated with low-dose anakinra while others received no anakinra. It’s unclear how this treatment decision was made. Patients treated with low-dose anakinra appeared to do better (with a 7-day survival of 100%), raising a question of whether low-dose anakinra might have been allocated to patients who were deemed more capable of recovering. This selection bias might conceivably have created a scenario where the standard treatment group had a worse prognosis, leading it to fare poorly when compared to the high-dose anakinra group.
- Several authors had conflicts of interest.
- Use of hydroxychloroquine and lopinavir/ritonavir may have affected the results.
Strengths of the study
- These authors performed useful research in the midst of a hospital under surge conditions, which is truly impressive.
- Hyperinflammation was defined in a simple and clinically useful way, using C-reactive protein and ferritin (rather than, for example, IL-6 levels). This may facilitate future research and clinical application, given the widespread availability of these tests.
- To date, most guidelines have focused on the use of anti-inflammatory therapies for patients with ARDS who have already been intubated. These authors have shifted the focus towards patients with severe hypoxemia who aren’t intubated, with a view towards avoiding intubation. Intervening earlier in the disease process seems sensible, especially given the iatrogenic harms associated with intubation.
- Definition of a potentially effective dose of anakinra is useful. This may help set the stage for further trials.

- This is a before/after study evaluating the use of anakinra for patients with COVID-19, severe hypoxemia, and hyperinflammation. The study included patients on noninvasive ventilation, with a view towards preventing intubation.
- Standard treatment doses of anakinra (100 mcg subcutaneously twice daily) were ineffective in decreasing the C-reactive protein level, so this dose was abandoned. However, high-dose anakinra (5 mg/kg intravenously twice daily) did correlate with improvements in mortality.
- Numerous design limitations prevent this study from establishing causality. However, the study may be useful in determining an optimal dose and time window for intervention, in preparation for subsequent trials.
- High-dose anakinra therapy involves administration of 5 mg/kg IV BID until clinical improvement (roughly one week). That’s a very large cumulative dose of anakinra. The anakinra supply in the United States is currently limited, which could make it logistically impossible to apply anakinra at many hospitals.
related
- IBCC section on cytokine storm
- Understanding sepsis-HLH overlap syndrome (PulmCrit, 2016)
Image credit: Photo by Evan Wise on Unsplash
references
- 1.Cavalli G, De Luca G, Campochiaro C, et al. Interleukin-1 blockade with high-dose anakinra in patients with COVID-19, acute respiratory distress syndrome, and hyperinflammation: a retrospective cohort study. The Lancet Rheumatology. May 2020. doi:10.1016/s2665-9913(20)30127-2
- 2.Shakoory B, Carcillo J, Chatham W, et al. Interleukin-1 Receptor Blockade Is Associated With Reduced Mortality in Sepsis Patients With Features of Macrophage Activation Syndrome: Reanalysis of a Prior Phase III Trial. Crit Care Med. 2016;44(2):275-281. doi:10.1097/CCM.0000000000001402
- Pulmcrit wee: The cutoff razor - April 15, 2024
- PulmCrit Blogitorial – Use of ECGs for management of (sub)massive PE - March 24, 2024
- PulmCrit Wee: Propofol induced eyelid opening apraxia – the struggle is real - March 20, 2024
Even tough anti-inflammatory drugs my be of benefit in COVID-10 for some patients, one shouldn’t forget that there is limited data about safety and adverse events. We experienced in our ICU clinical deterioration in two patients treated with Tocilizumab, one of them died. To our experience, we cannot rule out exacerbation of a cytokine storm after treatment with Tocilizumab, since the clinical worsening (coagulopathy, respiratory failure, shock, perimyocarditis) increased rapidly shortly after administration in both of these patients. We submitted a case report in a German journal (under review) and posted our experience as a preprint on researchsquare. Another preprint… Read more »
COVID-19, of course; luckily, there wasn’t COVID in 2010 😉
Important limit of the study:
Almost 10 years difference between standard treatment vs high dose anakinra treatment
I’m reading it as almost ten DAYS difference ….
Years of age, I guess
very nice review. your work during this pandemic has been amazing Josh.
and in the group discussions, (eg, the forum on peep and venting recently), I appreciate when they say, “what do you think, Josh?” and there’s a pause, breath, and your your measured thoughtful responses.
thank you, boss.
tom
merced
Excellent review as always, Josh. One thing you may want to update is the dose of the low dose anakinra group. Patients in this cohort received 100 mg of anakinra twice daily, not 100 mcg .
KM curve divergent from day one… immortal time bias?