CONTENTS
- Rapid Reference 🚀
- Diagnosis
- Naloxone basics
- Treatment overview
- Additional treatment may be needed for certain agents:
- Podcast
- Questions & discussion
- Pitfalls

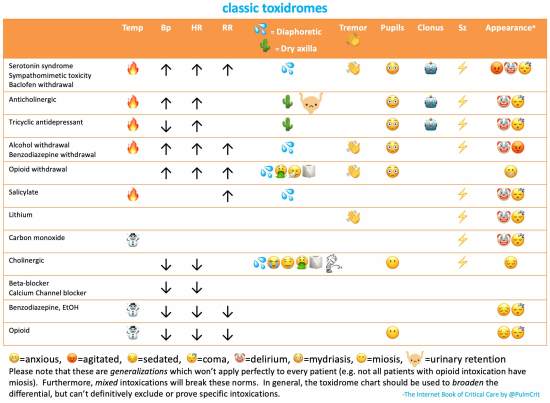
opioid toxidrome – key features
- Somnolence
- Reduced respiratory rate
- This is the most important clinical finding.
- Respiratory rate <12/minute was highly predictive of a response to naloxone in one series of patients presenting with altered mental status.(1996818)
- A normal respiratory rate or tachypnea suggests the absence of a clinically significant opioid toxidrome.
- 💡 Don't trust the recorded vitals, which are often incorrect. Count the patient's breaths manually.
- Pupil size?
- Classically, opioid intoxication causes small (“pinpoint”) pupils. However, severe opioid intoxication causes hypercapnia, triggering a sympathetic response which may normalize the pupil size.(25068603) Also, some opioids have serotonergic effects that dilate the pupils (e.g., meperidine and tramadol). Finally, coingestion with sympathomimetics may block this effect. Thus, small pupils suggest opioid intoxication, but normal pupil size doesn't exclude it.
“opioid-plus” syndromes
- Serotonin syndrome and seizures can be caused by meperidine, tramadol, or dextromethorphan.
- Methadone commonly causes QT prolongation and torsade de pointes.
- Loperamide in massive quantities may cause QT prolongation and torsade de pointes, but it may also cause QRS prolongation and monomorphic ventricular tachycardia (more on this below).
physical examination
- In addition to the usual toxicology examination, carefully examine the skin for any fentanyl patches.
intoxication with a different agent
- Alpha-agonist intoxication (e.g., clonidine) may cause bradycardia and somnolence, but without bradypnea.
- Benzodiazepine or alcohol ingestion may present similarly, with suppression of respiration and mental status.
- Gamma hydroxybutyrate (GHB) intoxication.
polysubstance intoxication
- Opioids are often coingested with acetaminophen when patients overdose on combination pills (e.g., Percocet or Vicodin).
- Opioids may be coingested with a sympathomimetic such as cocaine or amphetamine (a combination known as a “speedball”). In this case, administration of naloxone will unmask the effects of the sympathomimetic intoxication, often leading to marked agitation.
chronic opioid + acute pathology
- Many patients are on chronic opioids (either therapeutically or recreationally). The superposition of an acute illness (e.g., stroke) may create a confusing picture:
- Patients will often have evidence of opioid ingestion.
- Patients generally have a partial response to naloxone (e.g., with pupil dilation and agitation), but often without complete normalization of mental status. Increased arousal following naloxone is due to elicitation of opioid withdrawal, rather than reversal of opioid intoxication.
- The key to the diagnosis may be features which don't otherwise fit with an opioid toxidrome (e.g., fever, focal neurological findings, failure to improve over time, incomplete response to naloxone).
More on the differential diagnosis of delirium, or stupor/coma.
The evaluation should be tailored to the individual patient's presentation. Commonly involved:
- Acetaminophen & salicylate levels.
- Creatine kinase, if there is a concern regarding compartment syndrome from prolonged immobilization.
- Serum ethanol level.
- EKG.

naloxone basics
- Naloxone is an opioid antagonist.
- Administration of naloxone to someone not on opioids will have minimal effect (even in massive doses).
- For an acute opioid intoxication in a patient not chronically on opioids, large doses of naloxone are safe to use. The goal here is to fully block any opioid effect. If an excessive dose of naloxone is used, it won't matter. Consequently, for an acute opioid intoxication that is severe (e.g., procedural sedation gone awry with subsequent apnea), rapid administration of high doses of naloxone is logical.
- For patients who are on chronic opioids, administering high doses of naloxone may induce an immediate state of opioid withdrawal (including severe pain, agitation, and emesis). Consequently, for an acute-on-chronic opioid intoxication, the goal is to use just enough naloxone to prevent aspiration or apnea, but not so much as to induce withdrawal.
naloxone pharmacology
- Naloxone can be given via several routes (e.g., nebulized, intramuscular, or intravenous).
- Intravenous administration is preferred, if access is available.
- For emergent application in the emergency department, intramuscular access may be used.
- Following IV administration, the onset of action is prompt (within 1-2 minutes). This allows for rapid dose up-titration in situations where the patient isn't crashing.
- Naloxone's duration of action is about 20-90 minutes. Most opioids have a longer half-life than the naloxone, so the naloxone will wear off before the opioid intoxication resolves. Consequently, patients will often become re-sedated as the naloxone wears off.
naloxone up-titration
- For a patient who isn't in acute danger from apnea, naloxone can be gradually up-titrated (unless, as discussed above, the context is known to be an acute intoxication, where one can be much more aggressive in terms of up-titrating the dose).
- The starting dose is somewhat controversial, but beginning with ~0.05 – 0.1 mg is generally reasonable. Some advocate for using even smaller doses, which is entirely fine if there is sufficient time to dilute out the naloxone.
- The goal of naloxone administration is to achieve adequate ventilation and airway protection (not necessarily a full state of arousal). Shooting for airway protection may avoid eliciting opioid withdrawal, pain, and agitation.
- If naloxone isn't working, successively larger doses may be given every 3-5 minutes (e.g., 0.1 mg, 0.2 mg, 0.4 mg, 1 mg, etc.)
- The maximal naloxone dose which could be given is unclear. Many textbooks and articles refer to a maximal cumulative dose of 10 mg total. Patients who have large intoxications with synthetic fentanyl derivatives (e.g., carfentanill) might require higher doses of naloxone than have traditionally been used for heroin intoxication.
naloxone infusion
- Most opioids have a longer duration of action than naloxone, so repeated doses or an infusion of naloxone are often needed to prevent re-sedation.
- The usual starting dose for an infusion is ~2/3rds of the initial cumulative dose which was required to revive the patient (e.g., if the patient responds well to 1 mg, then start the infusion at 0.6 mg/hour). One or two PRN doses of naloxone may be needed while waiting to receive the naloxone infusion from pharmacy.
- For patients on chronic opioids, as their acute intoxication wears off the naloxone will start to induce a state of withdrawal. Patients will start complaining of pain, at which point the naloxone may be down-titrated or held (while observing the patient for possible re-sedation).
monitoring & serial assessment
- Respiratory rate:
- Perhaps the most useful parameter to trend.
- A respiratory rate falling below roughly 8-10 breaths/minute is concerning for hypoventilation.
- Oxygenation:
- Deteriorating oxygenation is highly worrisome, as these patients should oxygenate well once given naloxone.
- Chest x-ray may be needed to evaluate for aspiration (common) or opioid-induced noncardiogenic pulmonary edema (rare).
- Mental status & airway protection:
- Ideally, patients should be easily arousable and able to protect their airway.
- etCO2:
- If available, a nasal cannula capable of measuring end-tidal CO2 may be attached and used to trend the patient's CO2.
- Note, however, that these patients still require close attention to the other parameters listed above.
- Mild hypercapnia is acceptable as long as the hypercapnia is roughly stable and the patient is clinically arousable and protecting their airway.
Based on this assessment, patients can be categorized into five groups:

description of these patients
- These patients are not be exposed chronically to opioid.
- The most common scenario here is a patient who is administered opioid for procedural sedation (e.g., bronchoscopy).
- Since the patient does not chronically use opioid, there is no risk of provoking opioid withdrawal.
- Naloxone administration may unmask pain which is being treated by the opioid, but in many cases this is not a major consideration (e.g., bronchoscopy or colonoscopy procedures where discomfort is transient).
treatment
- Since there is no risk of inducing withdrawal, naloxone administration may be relatively aggressive.
- If there is an imminent threat of airway loss or hypoxemia, a large dose of naloxone should be immediately administered (e.g., ~4 mg IV).
description of these patients
- These patients are doing OK: they are protecting their airway, oxygenating, and ventilating.
- Patients may be sleepy, but are easily arousable.
- Respiratory rate should be reasonable (e.g., over roughly 12 breaths/minute).
- Oxygen saturation is adequate without supplemental oxygen.
treatment
- These patients generally do fine; they just need to sleep off the opioid.
- Careful observation is needed, with a focus on respiratory rate and airway protection.
- If deterioration occurs, transition to the “Too Sleepy Pathway.”
description of these patients
- They are oxygenating OK, although perhaps with the use of supplemental oxygen (the requirement for oxygen is concerning because this suggests that the patient may be hypoventilating).
- These patients are usually obtunded, with questionable ability to protect their airway.
- Their respiratory rate is low (e.g., respiratory rate below ~8-10 breaths/minute).
- Nonetheless, they are relatively stable (for the moment) and not actively deteriorating.
goals of naloxone treatment for these patients
- Diagnostic goal: clinical response indicates exposure to opioid.
- ⚠️ Caution: a patient on chronic opioids will respond to naloxone with increased alertness/pain, even if they aren't intoxicated with opioids. Thus, a partial response to naloxone proves opioid exposure, but doesn't exclude another process.
- Therapeutic goal: avoidance of intubation.
- The goal here is to give just enough naloxone so that the patient protects their airway. The therapeutic target is thus a patient who is comfortable and sleepy, but easily awakens and protects their airway.
- The goal isn't to bring the patient back to a normal state of consciousness. For patients on chronic opioids, trying to “normalize” their mental status may provoke pain and severe agitation.
initial naloxone dose
- Start at 0.05-0.1 mg IV, repeat dose every 2 minutes and gradually escalate (e.g., 0.05 mg, 0.1 mg, 0.1 mg, 0.2 mg, 0.2 mg, 0.4 mg, etc.).
- If there is a clinical response:
- 🛑 Stop as soon as the patient improves to a point where they are protecting their airway and ventilating adequately (e.g., respiratory rate >12/minute).
- If there is no clinical response:
- Most patients will respond to doses of naloxone below ~2 mg. However, some opioids could require higher doses (e.g., 10 mg).
- Overshooting?
- If you give too much naloxone too quickly, a patient on chronic opioid may develop pain and agitation.
- In this situation, the best approach is often to wait for naloxone to wear off, which should occur within an hour. If necessary, a small pain dose of ketamine may be used (e.g., 0.2 mg/kg). Try to avoid administration of opioid, as this leads to an ongoing vicious spiral.
redosing & naloxone infusion
- Naloxone lasts 20-90 minutes, so it will almost always wear off before the opioid intoxication resolves (causing the patient to revert into an opioid-intoxicated state).
- Many patients will require a naloxone infusion, which should be started at a rate equal to 2/3 of the initial loading dose per hour.
- Getting the infusion from pharmacy often takes 1-2 hours. Until the infusion is running, the patient will require intermittent doses of naloxone (e.g., repeating 2/3 of the initial dose hourly).
- As the opioid intoxication starts improving, the patient will become increasingly alert and a bit agitated. At this point, the naloxone infusion may be stopped or weaned (with close monitoring for possible re-sedation).
description of these patients
- These patients may be apneic or on the verge of complete respiratory arrest.
- Bag-mask ventilation is often required to maintain oxygenation and ventilation.
- Airway adjuncts (e.g., nasal trumpet) may be required to open the upper airway.
step #1: airway maneuvers & naloxone & preparation for intubation
- Basic airway management:
- The first intervention is to open the airway and perform bag-mask ventilation. Nasopharyngeal or oropharyngeal airways may be used to open the airway.
- If bag-mask ventilation isn't successful, move immediately to placement of a laryngeal mask airway (LMA).
- Naloxone
- This should be given simultaneously with airway maneuvers.
- Start with a high dose of naloxone (e.g., perhaps 4 mg IV or IM bolus).
- Escalate naloxone doses rapidly, up to a total of ~10 mg (especially if basic airway maneuvers are not working).
- Preparation for intubation
- This preparation should also occur simultaneously with the above interventions.
step #2a: intubation
- Intubation is indicated if either of the following occur:
- (a) Basic airway maneuvers fail to achieve adequate ventilation and the patient doesn't immediately respond to naloxone.
- (b) Basic airway maneuvers succeed in achieving adequate ventilation, which buys you some time. However, despite escalating doses of naloxone, the patient fails to awaken.
- Subsequently, proceed down the intubated pathway.
step #2b: if the patient responds to naloxone
- If the patient awakens following naloxone administration, then the need for intubation is averted.
- Proceed down the too-sleepy pathway.
- 🛑 Note that there is a high risk of re-sedation within the next 1-2 hours, so this patient remains in critical condition.
initial management
- For the intubated patient, there is generally no immediate need for naloxone.
- Initial management should focus on stabilization and exclusion of other problems (e.g., head CT scan, toxicology evaluation).
- Avoid use of any long-acting sedative (the best sedation strategy here is usually just propofol). If the patient starts requiring any significant dose of sedation, consider whether they may be extubated.
naloxone infusion to facilitate extubation
- For patients who fail to metabolize their opioid after 6-12 hours, it might be reasonable to perform a very gentle naloxone titration. If the patient responds well to a naloxone infusion, they may be extubated and continued on a naloxone infusion. Earlier extubation may avoid complications associated with mechanical ventilation.
- Candidates for this strategy should meet the following criteria:
- Uncomplicated opioid intoxication with a long-acting agent.
- No active lung disease (e.g., chest X-ray shows no aspiration).
- Any tests which may be desired (e.g., LP, MRI) have already been done.
notable properties of methadone
- Methadone has a long half-life, so it may take a while to wear off.
- Some patients may be on chronic methadone. Such patients may be at risk of withdrawal.
- Methadone increases the QT interval and can promote torsade de pointes.
treatment
- Follow the QT interval with serial EKGs.
- For isolated QT prolongation, monitor the patient on telemetry. Aggressively replete electrolytes to target a magnesium level >3 mg/dL and a potassium level >4 mEq/L.
- If torsade de pointes occurs, consider initiation of a magnesium infusion (more on the treatment of torsade de pointes here).
- For patients on chronic methadone maintenance therapy, consider consultation with a specialist in opioid use disorders to determine the best management. In general, patients may require ongoing methadone therapy to prevent withdrawal. An alternative course of action might be to transition to buprenorphine.
notable properties of loperamide
- Loperamide is an antidiarrheal agent, which may exert opioid effects when taken in large quantities. It may be used as a drug of abuse, or for the purpose of attempted detoxification from another opioid.
- Loperamide blocks hERG potassium channels, potentially causing a prolongation of the QT interval and torsade de pointes.
- This seems to be the primary cardiac toxicity of loperamide.
- Loperamide may also block sodium channels, causing a prolongation of the QRS interval that can lead to monomorphic ventricular tachycardia.
management
- (1) If there is an opioid toxidrome, this should be treated as per usual (e.g., with naloxone).
- (2) Management of potassium channel blockade:
- This is essentially the same as the treatment of methadone above: start with aggressive repletion of potassium and magnesium. If torsade de pointes occurs, manage as described here.
- (3) Management of sodium channel blockade:
- Management of sodium channel blockade is described in detail here.
- If the QRS interval is substantially prolonged, then initial management begins with administration of hypertonic bicarbonate boluses. If bicarbonate causes an improvement in the EKG, this confirms the presence of clinical sodium channel blockade. A maintenance bicarbonate infusion may subsequently be initiated. Be careful about hypokalemia induced by the bicarbonate infusion, as this may predispose to torsade de pointes (see #1 above).
- In cardiovascular collapse unresponsive to other measures, lipid emulsion may have a role.(31684751)
Follow us on iTunes
To keep this page small and fast, questions & discussion about this post can be found on another page here.

- BiPAP is NOT an appropriate treatment for opioid intoxication. Many people believe that any patient with hypercapnia requires BiPAP, which is wrong. A patient with opioid intoxication should be treated with naloxone as described above. If the patient is unable to ventilate adequately following naloxone, they should be intubated.
- Avoid excessive dosing of naloxone in a patient with chronic opioid exposure. The goal of naloxone is to arouse the patient sufficiently to protect their airway. The goal of naloxone is not to fully reverse the opioid intoxication, nor is the goal to normalize the blood gas values.
- Patients may look deceptively fine after getting naloxone, only to revert to a completely comatose state an hour later. This may cause problems during handoffs (e.g., the patient nearly stops breathing in the emergency department and is given naloxone, the admitting team evaluates the patient and thinks everything is fine, the patient is admitted and then rapidly reverts to a comatose state).
Guide to emoji hyperlinks 
= Link to online calculator.
= Link to Medscape monograph about a drug.
= Link to IBCC section about a drug.
= Link to IBCC section covering that topic.
= Link to FOAMed site with related information.
= Link to supplemental media.
References
- 01996818 Hoffman JR, Schriger DL, Luo JS. The empiric use of naloxone in patients with altered mental status: a reappraisal. Ann Emerg Med. 1991 Mar;20(3):246-52. doi: 10.1016/s0196-0644(05)80933-3 [PubMed]
- 25068603 Rollins MD, Feiner JR, Lee JM, Shah S, Larson M. Pupillary effects of high-dose opioid quantified with infrared pupillometry. Anesthesiology. 2014 Nov;121(5):1037-44. doi: 10.1097/ALN.0000000000000384 [PubMed]
- 31684751 Eggleston W, Palmer R, Dubé PA, Thornton S, Stolbach A, Calello DP, Marraffa JM. Loperamide toxicity: recommendations for patient monitoring and management. Clin Toxicol (Phila). 2020 May;58(5):355-359. doi: 10.1080/15563650.2019.1681443 [PubMed]