CONTENTS
- Rapid Reference 🚀
- Physiology
- Causes of ICP elevation
- Diagnosis
- ICP monitoring
- Treatment
- Podcast
- Questions & discussion
- Pitfalls
Monro-Kellie doctrine
- The volume within the skull is fixed. This volume is composed of brain tissue (~80%), blood volume (~10%), and CSF volume (~10%).
- The Monro-Kellie doctrine states that any increase in the volume of one component must lead to a reduction of the volume of one or both of the remaining two components. This is essentially a statement regarding the conservation of volume within the skull.
compliance curve of the brain
- The relationship between volume and pressure within the brain may be understood roughly as shown in the compliance curve below.
- Initially, changes in volume cause relatively little increase in pressure (e.g., due to displacement of blood from the venous sinuses, and of CSF out of the skull).
- Beyond a certain point, the pressure will rapidly increase.
- The clinical significance of this is that in some situations, relatively small shifts in intracerebral volume may cause large differences in the intracranial pressure.
- The compliance curve may be steeper in younger patients, who have no cerebral atrophy and thus little room to accommodate swelling.

CSF is synthesized in the choroid plexus within the cerebral ventricles, at a surprisingly high rate (~20 ml/hour). CSF subsequently passes through the brain's ventricular system, wraps around through the subarachnoid space, and is reabsorbed by the arachnoid granulations near the apex of the skull (figure below). Abnormalities in this process may give rise to two types of hydrocephalus.
communicating hydrocephalus
- Basic physiology: There is an overall imbalance between CSF production and CSF reabsorption. This leads to elevation of pressure throughout the brain's ventricular system and subarachnoid space.
- Causes:
- Inadequate removal of CSF by the arachnoid granulations (e.g., due to meningitis, subarachnoid hemorrhage, leptomeningeal carcinomatosis, or thrombosis of the superior sagittal sinus).
- Excessive production of CSF due to a papilloma of the choroid plexus.
- Imaging findings: Initially, communicating hydrocephalus may be more difficult to diagnose based on CT scans, because it doesn't immediately cause large tissue shifts. Findings may include:
- (1) Symmetrical expansion of all ventricles.
- Blunting of the frontal horns of the lateral ventricles may be a useful sign.
- The third ventricle may appear dilated and rounded (rather than it's usual slit-like configuration).
- (2) Effacement of cerebral sulci and compression of cisterns may occur.
- (1) Symmetrical expansion of all ventricles.
- Management: Communicating hydrocephalus may be treated by removal of CSF from anywhere (e.g., a lumbar drain or an intraventricular drain).
noncommunicating (or obstructive) hydrocephalus
- Basics: An obstruction within the ventricular system blocks the flow of CSF, leading to a localized upstream backup of CSF. For example, obstruction of the fourth ventricle will cause CSF to accumulate in the lateral and third ventricles. Alternatively, a single lateral ventricle may become obstructed (“trapped”), causing it to expand.
- Causes:
- Obstruction due to intraventricular hemorrhage or inflammation, often occurring at the fourth ventricular foramen or the cerebral aqueduct.(26046515)
- Mass lesion which directly blocks the flow of CSF or indirectly compresses the CSF drainage system.
- Imaging findings:
- Noncommunicating hydrocephalus generates pressure differentials within the brain. This leads to tissue shifts and herniation. Noncommunicating hydrocephalus is therefore easier to diagnose based on CT scans, because pressure differentials will cause shifts in brain tissue visible on CT.
- Acute obstructive hydrocephalus may cause CSF to leak from the ventricles into the brain tissue, producing interstitial edema surrounding the ventricles (which may be more notable on MRI than on CT).(26046515)
- Management: Noncommunicating hydrocephalus must be treated by removal of CSF from a location upstream of the obstruction (e.g., typically an external ventricular drain that removes CSF from a lateral ventricle). In contrast, removing CSF from a location downstream of the obstruction (e.g., via a lumbar puncture) could precipitate herniation by increasing pressure gradients within the brain.
Understanding types of edema is a fundamental concept in neurocritical care.
vasogenic edema
- Pathophysiology: Due to disruption of the blood-brain barrier, with leakage of fluid out of capillaries.
- Diagnostics:
- Vasogenic edema often has a tendency to involve the white matter, while sparing the grey matter (thus preserving the grey-white junction). Mass effect may occur.
- CT scan: Hypoattenuation.
- MRI:
- Hyperintensity on T2/FLAIR, without diffusion restriction.
- Contrast enhancement often occurs due to loss of the blood-brain barrier.
- Lumbar puncture may show elevated CSF protein due to increased endothelial permeability. (Louis 2012)
- Causes include the following: (*indicates processes that may present on imaging predominantly with vasogenic edema):
- Tumors (there is often a rim of edema surrounding the tumor itself).
- Infection (e.g., abscess, encephalitis*).
- Inflammation (e.g., demyelinating diseases*).
- Traumatic tissue damage, hemorrhage.
- Cerebral venous thrombosis.*
- Posterior reversible encephalopathy syndrome.*
- Management:
- Many causes of vasogenic are responsive to steroid (e.g., edema due to tumors, infection, or inflammation).
- Efficacy of osmotherapy may be limited by blood-brain disruption. (Louis 2012)
cytotoxic edema
- Pathophysiology:
- Neuronal injury causes swelling of neurons. Permanent neuronal cell death often occurs, but not invariably.
- Less often, edema of the myelin sheaths of neurons (intramyelinic edema) can cause diffusion restriction that is reversible.(31589567)
- Imaging:
- Affects both grey and white matter.
- CT: Hypoattenuation, may cause loss of grey/white junction.
- MRI:
- Key finding is diffusion restriction, which leads to hyperintensity on DWI sequences and hypointensity on ADC sequences.
- Increased signal on T2/FLAIR may occur secondarily.
- Causes include:
- Focal ischemic stroke is the most notable cause.
- Global hypoxic-ischemic brain injury.
- Acute hepatic failure.
- Prolonged seizures.
- Management: Cytotoxic edema is not responsive to steroid.
interstitial edema (aka hydrostatic edema)
- Pathophysiology: Obstructive hydrocephalus causes CSF to infiltrate the periventricular white matter.
- Imaging features: hydrocephalus with edema surrounding the ventricles.
- Management: treatment of hydrocephalus (e.g., external ventricular drain).
osmotic cerebral edema
- Occurs when an osmotic gradient favors entry of water into the brain. May be more likely to happen if preexisting vasogenic or cytotoxic edema challenge the brain's ability to regulate volume in the face of an osmotic challenge.(34618757)
- Causes include:
- Acute hyponatremia.
- Dialysis disequilibrium syndrome.
- Rebound edema (following rapid weaning of hyperosmolar therapy).
- Imaging features: The entire brain volume expands, which may be difficult to notice.
ICP may be elevated by a focal lesion, or global tissue swelling. Although there is often overlap between these situations, it's worth considering some differences between these extremes:
focal lesion (e.g., epidural hematoma)
- Pressure may be elevated locally, but not diffusely throughout the brain.
- Nearby tissue may be at highest risk (e.g., due to herniation).
- CT scan will easily identify the abnormality (based on local tissue shifts).
- An EVD (external ventricular drain) may be less useful, since pressure may not be elevated uniformly throughout the entire brain.
diffuse tissue edema (e.g., acute liver failure)
- Pressure is elevated throughout the entire brain, in a more diffuse fashion.
- A primary concern is inadequate global perfusion, due to low cerebral perfusion pressure.
- CT scan may be less sensitive to detecting the abnormality (if the entire brain swells, without causing tissue shifts).
- An EVD (external ventricular drain) may be more useful, since pressure is elevated throughout the entire brain.
intracranial mass lesion
- Tumor (primary or metastatic).
- Hematoma (e.g., epidural, subdural, intraparenchymal).
- Abscess or subdural empyema.
- (Note that mass lesions may occasionally cause obstructive hydrocephalus as well as local mass effect.)
cerebral edema (diffuse edema of brain tissue)
- Focal edema
- Large ischemic stroke.
- Perihematomal edema.
- Global edema
- Traumatic brain injury with diffuse axonal injury.
- Anoxic brain injury.
- Hyperammonemia of any etiology, for example:
- Fulminant hepatic failure.
- Urea cycle abnormalities.
- Hyponatremia (severe and acute).
- Hypercapnia.
- Hyperthermia.
- Severe encephalitis.
- PRES (posterior reversible encephalopathy syndrome).
- Post-carotid endarterectomy hyperperfusion syndrome.
communicating hydrocephalus: impaired CSF reabsorption
- Meningitis:
- Bacterial meningitis.
- Cryptococcal meningitis.
- Leptomeningeal carcinomatosis.
- Subarachnoid hemorrhage.
- Thrombosis of the superior sagittal sinus.
- (Rarely may be caused by excessive CSF production due to a papilloma of the choroid plexus.)
obstructive hydrocephalus
- Any lesion obstructing the flow of CSF (most often at the fourth ventricular foramen or cerebral aqueduct).
- Any parenchymal mass lesion causing extrinsic compression of the ventricular system.
- Intraventricular obstructive lesion (e.g., colloid cyst).
obstruction of venous outflow from the brain
- Cerebral venous sinus thrombosis (especially involving the sagittal sinus).
- Jugular vein compression or thrombosis.
- External neck compression.
- Superior vena cava syndrome (SVC syndrome).
- Severe systemic venous congestion.
- Thoracic or intra-abdominal compartment syndromes (more on this below)
clinical findings which may occur with diffuse elevation of intracranial pressure
- Headache (exacerbated by coughing, sneezing, lying down, or exertion).(32440802)
- Vomiting (often not precipitated by nausea, may be projectile vomiting, and may be refractory to medical therapy).
- Altered consciousness (this may be a late or variable feature; usually somnolence occurs, but agitation is also possible).
- Visual alterations (vision loss, flashing lights, diplopia).
- Abducens nerve palsies (dysfunction of cranial nerve 6, causing inability to gaze laterally).
- Cushing's triad (hypertension, bradycardia, irregular respirations)
- The triad is usually a late manifestation of severe brain injury, but isolated hypertension may be seen early.
herniation syndromes
- Focal elevation of intracranial pressure may lead to herniation.
- More on herniation syndromes: 📖
5-Minute Sono by Dr. Jacob Avila
- Optic nerve sheath diameter may be measured with ultrasound as shown in the video above.
- When measured 3 mm behind the eye:
- <5 mm is normal.
- 5-6 mm is a grey zone.
- >6 mm suggests abnormal ICP.
- Ultrasonographic papilledema may also be detected with the same exact technique.
- This is supported by less evidence compared to optic nerve sheath diameter, but it appears to have good performance.
- Papilledema may be less accurate for immediate-onset intracranial hypertension, because it takes some time to develop.
- Papilledema may be useful for identifying ICP elevation in cases where the nerve sheath is borderline (e.g., between 5-6 mm wide). More on this here.

- The performance of CT scan varies depending on the cause of ICP elevation. CT is fantastic for focal anatomic lesions. In this situation, edema will cause tissue shifts which are easily seen.
- Some patients have elevated intracranial pressure with an anatomically normal brain (e.g., if due to meningitis, hepatic failure, pseudotumor cerebri). In these situations, ICP may be elevated despite a normal CT scan.
- 🛑 Normal CT imaging doesn't exclude elevation of intracranial pressure.
lumbar puncture opening pressure reflects ICP
- For patients who are lying in a supine position, the opening pressure of the lumbar puncture is a validated measurement of intracranial pressure.(17210899, 9115655)
- Note that pressure is initially measured in cm of water. This needs to be multiplied by 0.7 to convert to mm of mercury.
clinical roles of measuring ICP via lumbar puncture
- In the presence of focal pathology, lumbar puncture may be dangerous. This is particularly true in the case of noncommunicating hydrocephalus or mass lesions with threatened downward herniation.(as discussed above)
- In the absence of a mass lesion or CSF obstruction, lumbar puncture is a safe strategy to measure intracranial pressure. This may be both diagnostic and therapeutic, since removal of CSF via lumbar puncture will be beneficial among patients with communicating hydrocephalus.
- Examples of pathologies where lumbar puncture may be useful as a diagnostic tool to reveal elevated ICP include meningitis and pseudotumor cerebri. More discussion on the role of CSF removal in meningitis here.
general indications for ICP monitoring
- General indications to consider ICP monitoring:
- (1) Patient is known or extremely likely to have elevated ICP (e.g., based on CT scan showing significant intracranial mass effect).(Louis 2021)
- (2) Neurological examination is very poor, which doesn't allow for meaningful serial clinical neurological examinations.
- (3) Prognosis is favorable enough to merit aggressive care.
- For patients with hydrocephalus, and EVD (external ventricular drain) is often both diagnostic and therapeutic.
- The benefit of ICP monitoring may be greater in certain disease states (e.g., traumatic brain injury) as compared to others (e.g., ischemic stroke).
- There is no high-quality evidence that monitoring and treating ICP is beneficial. In one prospective RCT of patients with traumatic brain injury, ICP measurement didn't affect outcomes.(23234472) There is a risk that ICP monitoring could lead to overaggressive therapies and iatrogenic harm in the quest for a “normal” ICP value.
disease-specific indications:
- In TBI (traumatic brain injury 📖), ICP monitoring is generally recommended for patients with GCS≤8 and CT scans showing hematomas, contusions, swelling, herniation, or compressed basal cisterns.(27654000)
- In SAH (subarachnoid hemorrhage 📖), an external ventricular drain is generally indicated for patients with acute hydrocephalus.
external ventricular drain (EVD)
- A burr hole is used to penetrate the skull, after which a catheter is blindly advanced into the lateral ventricle using anatomic landmarks. The procedure may be performed at the bedside.
- This allows for continuous pressure transduction, CSF sampling, and therapeutic drainage of CSF.
- Therapeutic drainage of CSF provides definitive therapy for hydrocephalus.
- A ventricular drain allows for re-zeroing of the pressure transducer, ensuring there is no “drift” in values over time.
- This is the most accurate form of ICP monitoring, but also the most invasive. Risks include:
- The primary concern is generally ventriculitis, which occurs in ~10% of patients.
- The catheter is blindly inserted through brain tissue, which may be difficult in patients with compressed ventricles or significant midline shift (requiring multiple passes). There is a risk of causing hemorrhage during insertion through brain tissue.
- Among patients with compressed, slit-like ventricles it may be impossible to place an external ventricular drain.(Nelson, 2020)
- More on external ventricular drains here: 📖
intraparenchymal monitor
- A monitor is inserted into peripheral brain tissue for continuous pressure measurement.
- The main advantage is that this is less invasive than an external ventricular drain and easier to place, with a lower infection rate.
- Codman and Raumedic sensors are compatible with MRI, but Camino and Pressio sensors are not.(35816237)
- Drawbacks:
- (1) CSF cannot be sampled diagnostically, nor removed therapeutically.
- (2) Local pressure may not necessarily reflect pressure throughout the brain.(Louis 2021)
- (3) Over the course of a week, the pressure measurement tends to drift. Calibration is not possible following insertion. However, this may be less of a problem with fiberoptic devices (as opposed to strain gauge devices).(Louis 2021)
(subarachnoid bolt)
- This involves a hollow screw placed through the skull, communicating with the subarachnoid space.
- The advantage of this device is its low risk of infection or hemorrhage. Unfortunately, bolts often become clogged and nonfunctional.
- Subdural and epidural monitors are less accurate and rarely used in clinical practice.(35816237)
intracranial pressure (ICP)
- Normal ICP is 5-15 mm Hg (8-20 cm water).(Louis 2021; Flemming 2022)
- Levels >~20-22 mm Hg (>27-30 cm water) which are sustained for over ~10-15 minutes are often regarded as an indication to institute therapy to reduce the ICP (sometimes referred to as an “ICP crisis”).
- Transient ICP elevations due to coughing or bucking the ventilator should not trigger intervention. Treatment triggered by spontaneous ICP oscillations may lead to overtreatment of self-limited ICP oscillations.(30516597)
- ICP can fluctuate substantially over time, so it may be challenging to dichotomize a single value as either “normal” or “elevated.”
- When considering the ICP value, several factors may be considered in a holistic fashion:(35058406)
mm Hg vs. cm water
- 1 cm of water equals 0.74 mm of mercury (Hg).
- Considerable variability exists regarding which units are utilized.
- 96% of studies in the literature report pressures in terms of mm.(31773310)
- Standard and consistent reporting of ICP in terms of mm might be desirable. The use of mm facilitates immediate calculation of the cerebral perfusion pressure (which is equal to MAP-ICP).
- ⚠️ At most centers, the pressure is recorded in mm Hg, but the drip chamber height of an external ventricular drain is measured in cm water.(Albin 2022)
cerebral perfusion pressure (CPP)
CPP = (MAP – ICP)
- The cerebral perfusion pressure is the driving pressure for perfusion of blood through the brain.
- A normal CPP is roughly ~50-90 mm.(Jallo 2021)
- The CPP should ideally be maintained above roughly >60 mm.
- CPP is often the best surrogate which we have for estimating cerebral blood flow.
- 🛑 Don't get too focused on the ICP, while ignoring the CPP. Note that patients with pseudotumor cerebri can function well and have normal mental status despite severely elevated ICP levels.(31138948) Thus, an isolated elevation of ICP with a good clinical exam and adequate CPP isn't necessarily an emergency.
- Normally, cerebral autoregulation will maintain a stable cerebral blood flow across a wide range of CPP values (e.g., ~50-140 mm). However, an injured brain may lose autoregulatory capacity, causing blood flow to increase in response to increases in CPP. Thus, maintenance of an optimal CPP is increasingly important in the context of more severe neurologic injury.
- ⚠️ If an arterial catheter is being used to evaluate the CPP, it should ideally be zeroed at the tragus of the ear (so that the arterial catheter matches up with the external ventricular drain).(9187616,) If the ICP is zeroed based on the ear while the MAP is zeroed based on the heart, this may overestimate the CPP by up to >10 mm.(Louis 2021)

treatment: defending the cerebral perfusion pressure
- It's possible that achieving an adequate CPP may be more important than achieving a normal ICP (remember, in physiology everything depends on pressure gradients).
- CPP should ideally be maintained above >60 mm. If the CPP falls substantially below ~50-60 mm, this threatens the perfusion of brain parenchyma.
- Different texts disagree about the ideal CPP target. In reality this may vary between patients – patients with longstanding hypertension may require a higher CPP to achieve adequate brain perfusion.
- Elevation of the MAP with vasopressors may allow the CPP to be rapidly increased. Fluid administration is less effective at increasing the MAP, generally failing to achieve a sustained improvement in CPP.
- Typically, either norepinephrine or phenylephrine may be utilized. The choice may depend on hemodynamic specifics, including heart rate (norepinephrine will tend to increase the heart rate, whereas phenylephrine tends to decrease the heart rate).
- For patients with ICP elevation whose precise ICP is unknown, it may be reasonable to target a higher Bp than normal (e.g., target MAP >75-80 mm).
three components of the ICP waveform
- P1 = Percussion wave 🥁
- Generated by arterial pulsations at the beginning of systole.
- P2 = Tidal wave 🌊
- Reflects the compliance of the ventricles, responding to increased pressure of the percussion wave.
- Usually P2 is ~80% as tall as P1.(28169966) If P2 > P1 this suggests poor compliance, due to concerningly elevated ICP.
- P3 = Dicrotic wave 📉
- Reflects aortic valve closure at the end of systole.
waveforms in the context of elevated intracranial pressure
- Normally, P1 > P2 > P3, in a descending pattern.
- As ICP elevates, the brain compliance is compromised. This causes P2 to increase higher than P1. Eventually, P1 and P2 may fuse into a single wave (figure above).
- Abnormal waveforms may occur before the absolute value of the ICP is elevated. This may be an early signal of an impending ICP crisis.(34618757)
- Dampened waveforms may also be seen in patients with catheter occlusion, catheter malposition, air bubbles, cerebral vasospasm, or in patients with an open skull (e.g., status post craniectomy).(28169966; Greenberg 2016)
physiology

- Plateau waves result from a vicious cycle as shown above, which causes transient elevations in ICP.
- The physiological prerequisites for plateau waves include the following:
- (1) Tenuous brain perfusion.
- (2) The brain is on the steep, right portion of the cerebral compliance curve (see figure below). The brain has expanded to the point where there is really no extra room to accommodate further edema. Thus, the mere dilation of cerebral vasculature is sufficient to increase the ICP.
- (3) Preserved cerebral autoregulation.
- Plateau waves themselves are self-limiting. Short plateau waves (e.g., ~5-15 min) pose no immediate threat, but longer pressure waves might cause neurologic insult. Regardless, the presence of pressure waves reflects a dangerous situation, where the brain has reached the limits of physiological compensation.
clinical aspects

- Plateau waves (a.k.a. pathological A waves) are transient, periodic severe elevations in ICP (up to 50-100 mm) usually lasting for ~5-30 minutes (but potentially up to two hours).(19565359) They may occur spontaneously, or may be precipitated by coughing, pain, or changes in position from lying to sitting or from sitting to standing.(26704760)
- Plateau waves are often accompanied by clinical deterioration, with symptoms as listed below.(29329250) Arterial blood pressure remains roughly stable during a plateau wave, although dysautonomia can occur.(19565359)
- Pupillary dilation.
- Reduced consciousness.
- Ataxia, tonic posturing.
- Headaches, nausea/vomiting
- Visual loss.
- Plateau waves will generally be misdiagnosed as other phenomena:
- Plateau waves can mimic seizures (but this relationship is complicated because seizures may also trigger plateau waves).
- Since plateau waves may be triggered by standing, they can be confused with orthostatic hypotension.(26704760)
- Plateau waves will eventually resolve on their own. However, this still may be dangerous since episodes persisting for >30 minutes may cause irreversible damage due to impaired perfusion.(Nelson, 2020)
- Plateau waves may also be terminated by interventions that cause cerebral vasoconstriction (e.g., hyperventilation or vasopressors that induce hypertension).(24411105)
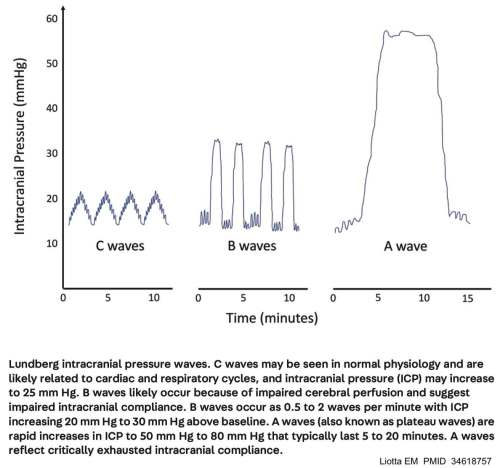
other Lundberg waves
- B waves:
- Waves last between ~30 seconds to several minutes.
- Amplitude is ~20-30 mm Hg.
- Significance is unclear; often suggest impaired intracranial compliance.
- C waves:
- Waves last ~7-15 seconds.
- Amplitude is usually <20 mm Hg.
- C-waves are a component of normal physiology, due to cardiac and respiratory cycles.(Albin 2022)
Treatment for ICP elevation can be loosely divided into three rough tiers, depending on how aggressive the treatments are (table below). Please note, however, that this is intended only as a rough cognitive rubric. It will often be appropriate to mix treatments from different tiers.
If possible, the best management of elevated ICP is definitive treatment of the causative disease process. All other strategies in the management of ICP are merely temporary approaches to support the patient, as a bridge to recovery.
Treatment will depend on the underlying disease. Example may include:
- Resection of a tumor.
- Management of metabolic disorders.
- Placement of a ventriculoperitoneal shunt may be necessary in some patients with refractory hydrocephalus.
- Steroid may be beneficial for some forms of vasogenic edema (more on vasogenic vs. cytotoxic edema above).
These constitute good practice for any patient with neurological injury (regardless of ICP). However, these practices may be especially important among patients with ICP elevation.
basic measures to avoid excess intrathoracic pressure
- Adequate analgesia and sedation should be provided, to avoid coughing or bucking the ventilator.
- Avoid excessively elevated airway pressures (e.g., unnecessarily high levels of PEEP).
- More on the modulation of intrathoracic pressure below.
avoid compression of the jugular veins
- Try to keep neck in a neutral position.
- Avoid using a C-collar (cervical collar).
- Avoid central lines in the internal jugular position, if possible.
normocapnia
- Decreasing PaCO2 causes vasoconstriction of cerebral arteries. This may reduce ICP, but it also reduces cerebral blood flow.
- For most patients with ICP elevation, a reasonable target is a low-normal PaCO2 (e.g., 35-40 mm).
- Carbon dioxide levels should be monitored carefully, typically using a combination of end tidal CO2 monitoring with intermittent blood gas measurement as needed.
- ⚠️ Hyperventilation will decrease the ICP, but it will also reduce cerebral perfusion by causing cerebral vasoconstriction. Hyperventilation should be reserved for use as a short-term bridge to immediate neurosurgical intervention (e.g., targeting a PaCO2 of ~25-30 mm). Prolonged periods of hyperventilation will lead to cerebral hypoperfusion, secondary ischemic injury, and subsequently worsening cerebral edema.
normonatremia
- Target a normal sodium level.
- The most important aspect is to avoid any large and rapid decreases in the sodium level. Chronic hyponatremia may be tolerated if the patient is otherwise doing well and the sodium is stable.
- Avoid hypotonic fluid infusions in patients who may have elevated ICP.
normothermia
- A fever increases ICP by increasing the brain’s metabolic demands and need for arterial blood supply.
- Patients with elevated ICP should be maintained at a normal temperature. In some cases this may be achievable with simple interventions (e.g., scheduled acetaminophen). Refractory fever can be managed with an external adaptive temperature control device.
- If there is difficulty reducing the patient's temperature, techniques described in the chapter on hyperthermia may be used.
head of bed elevation ??
- Elevation of the head of the bed is widely recommended, but without a robust evidentiary basis. Elevation of the head of the bed will tend to reduce the ICP – but this comes at the cost of reducing the arterial perfusion in the brain.
- One study found that elevating the head of the bed actually reduced the cerebral perfusion pressure and increased the incidence of plateau waves, indicating potential harm due to elevation.(3772451) Consequently, some advocate for patients to lie in a flat position, in order to maximize the cerebral perfusion pressure! Elevating the head of the bed >45 degrees in particular should be avoided as this may cause excessive reduction in the cerebral perfusion pressure.(32440802)
preferred agents for emergent therapy of ICP elevation
- Hypertonic bicarbonate ampules (typically 50 ml of 1 mEq/ml bicarbonate):
- This has the same tonicity as “6% saline” – making it roughly twice as potent as 3% saline.
- 2-3 ampules of hypertonic bicarbonate (100-150 ml) may be used as emergent therapy for elevated ICP.
- The advantage of this therapy is that it's generally immediately available.
- Hypertonic bicarbonate may be contraindicated in patients with a preexisting metabolic alkalosis, which will be exacerbated by the additional bicarbonate.
- More on hypertonic bicarbonate for management of ICP elevation: 🌊
- 3% saline:
- May be given as a bolus of 250 ml.(33896520)
- Extensive literature supports that it's safe to administer 3% saline via a peripheral line.
- 🛑 Placing a central line to facilitate infusion of 3% saline is not indicated and may be potentially harmful (especially if the line is placed in the jugular position).
- 23.4% saline:
- 23.4% saline may be given as a 30-ml bolus over 10 minutes.(31773291) Faster administration may risk heart failure due to volume overload.(34618757)
- 23.4% saline is ideally administered via a central venous catheter, but this may not be practically feasible. Intraosseous access may be used in emergencies.(34618757) One large series reported peripheral administration in 57 patients without adverse events.(34173156) Thus, administration of 23.4% saline via a well-functioning peripheral line is permissible if this is deemed necessary for neuroresuscitation.
- Hypertonic sodium acetate:
- Ongoing therapy with hypertonic saline will lead to a non-anion gap metabolic acidosis (NAGMA).
- Hypertonic sodium acetate is helpful for patients with NAGMA. This may increase the serum sodium, while simultaneously treating NAGMA.
- For patients who require ongoing hypertonic therapy, sometimes treatment may alternate between hypertonic saline (which causes NAGMA) and hypertonic sodium acetate (which treats NAGMA). If the chloride levels are rising while using NaCl, then consider switching to sodium acetate. One RCT found that using a balanced hypertonic solution that contained both NaCl and sodium acetate resulted in reduced rates of acute kidney injury, compared to hypertonic NaCl.(32391156)
monitoring hypertonic therapy for safety
- 👀 Avoid excessive hypernatremia. The sodium should be increased to no higher than ~155 mEq/L. (31659383, 32227294, 31659383)
- 👀 Avoid non-anion gap metabolic acidosis (NAGMA) and/or hyperchloremia (e.g., serum chloride >110-115 mEq/L).
- If NAGMA and/or hyperchloremia are developing, switch from NaCl to either hypertonic bicarbonate or sodium acetate.
- 👀 Avoid increasing the sodium by more than >6-8 mEq/day, to avoid osmotic demyelination syndrome.
- Avoid volume overload (e.g., with the use of diuretics to maintain an even fluid balance).
continuous infusions of hypertonic therapy probably don't help
- Continuous infusions of 3% saline are commonly employed, but they are not well supported by evidence. 🌊 Continuous exposure to hypertonic therapy merely causes the brain to adapt to a higher tonicity, thereby minimizing any benefit.
- Rather than a continuous infusion, a better strategy may be using intermittent boluses of hypertonic therapy, as clinically indicated.
- Everything that goes up must come down. Increasing the patient's sodium may cause a transient improvement in ICP, but eventually the sodium will need to be lowered back to normal. Unfortunately, the brain tissue will rapidly adapt to being hypernatremic, so lowering the sodium back to normal carries a risk of rebound ICP elevation.
- 🛑 If the patient's sodium has been elevated for more than 1-2 days, watch for rebound intracranial hypertension when hypertonic therapy is being discontinued. In this situation, it may be safer to taper off hypertonic therapy rather than to stop it abruptly, to reduce the risks of acute ICP elevation.
reasons to stop using mannitol
- 🛑 Mannitol is potentially nephrotoxic.
- 🛑 Mannitol often induces diuresis, which may lead to volume depletion and brain hypoperfusion. Diuresis may also promote electrolytic disarray (with hypokalemia, hypomagnesemia, and hypophosphatemia).(32440802) This risk can be mitigated by replacing the diuresed volume 1:1 with an isotonic solution (e.g., plasmalyte) for 6 hours after mannitol infusion.
- 🛑 Guidelines recommend monitoring the efficacy and safety of mannitol by following the serum osmolality to ensure that mannitol isn't accumulating and thereby increasing the risk of kidney injury (e.g., an osmolality >320 mOsm and/or osmolal gap >55 mOsm suggests mannitol accumulation).(32227294) In reality, this is rarely performed properly.(32440802)
- 🛑 There may be a rebound elevation in ICP, due to mannitol which crosses the blood-brain barrier and eventually causes edema within damaged areas of the brain.
- 🛑 Meta-analysis of available studies shows that mannitol is less effective than hypertonic saline, while being associated with higher rates of kidney injury.(21242790) A Cochrane analysis found that mannitol might have detrimental effects on mortality, when compared to hypertonic saline.(32440802)
- 🛑 Guidelines recommend the use of hypertonic saline rather than mannitol for traumatic brain injury or intracranial hemorrhage.(32227294)
avoidance of coughing or bucking the ventilator
- Coughing or ventilator dyssynchrony may increase intrathoracic pressures, thereby increasing the ICP.
- Overall, the sedation target for patients with elevated ICP may be a bit deeper than that of most patients.
- Coughing may be avoided by avoidance of suctioning, use of propofol, and addition of PRN opioids for breakthrough coughing.
- For patients with refractory coughing or ventilator dyssynchrony, temporary paralysis may be considered.
moderately deep sedation with propofol
- Propofol reduces brain metabolic activity, which may improve ICP and also avoid cellular hypoxemia.
- For patients with ICP elevation, propofol sedation may be a sensible choice. Additionally, it may be reasonable to target a slightly deeper level of sedation than might otherwise be chosen.
- Propofol has the advantage that it is easily titrated, thereby allowing for neurologic examinations to be performed intermittently. Propofol also has antiepileptic properties that will protect against seizure.
barbiturate coma
- The concept of a barbiturate coma is the same as that of propofol use above, with the difference that barbiturates can achieve a deeper coma. Barbiturates are ideally titrated using an EEG, to achieve a burst-suppression pattern (once a burst-suppression pattern is reached, barbiturate dose should not be increased further).(31659383)
- Pentobarbital is preferred over phenobarbital, because the half-life of pentobarbital is somewhat shorter. Nonetheless, pentobarbital still has a long half-life (15-50 hours). Thiopental might be an attractive option here, but it's unavailable in the United States, due to its history of being used for lethal injection.
- A deep barbiturate coma has substantial side effects. Hypotension requiring vasopressor support may occur, as well as ileus. Barbiturates are metabolized slowly, which may make it impossible to evaluate the patient's neurological outcome for weeks. This may lead to some very challenging situations. For example, it's ethically impermissible to withdraw life-sustaining therapy in a patient who has been rendered comatose with pentobarbital.
- ⚠️ Due to inability to titrate or rapidly reverse this therapy, barbiturate coma is an intervention of last resort.
- Nonconvulsive status epilepticus (NCSE) is a potential cause of elevated intracranial pressure.
- EEG should be considered to exclude nonconvulsive status epilepticus as a component of Tier 1-3 therapy.(27654000; 34618760)
- Seizure prophylaxis may be indicated in certain contexts, depending on the specific brain injury.
EVD (external ventricular drain) for hydrocephalus
- If hydrocephalus is the cause of ICP elevation, this can be treated effectively by placement of a ventricular drain (with therapeutic drainage of CSF).
decompressive hemicraniectomy
- Management of refractory elevation in ICP may involve a decompressive craniectomy (removal of part of the skull and opening of the dura, to allow the brain to swell).
- The utility of decompressive craniectomy depends on the underlying disease process and associated prognosis. Evidence is best for younger patients with large strokes involving the middle cerebral artery.
forced normothermia (36 C)
- Avoidance of fever should be achieved in any patient with ICP elevation.
- Scheduled acetaminophen is often adequate. If this fails, use of an external adaptive cooling system may achieve definitive temperature control.
induced hypothermia (33 C)
- Hypothermia does reduce the ICP. However, hypothermia may also cause bradycardia and hypotension – which may impair brain perfusion.
- There is no high-quality evidence that hypothermia improves clinical outcomes among patients with ICP elevation. The EUROTHERM trial found evidence of harm due to therapeutic hypothermia for management of ICP elevation due to traumatic brain injury. 🌊 (31773291)
transmission of pressure between compartments
- Increased intrathoracic pressure may be transmitted to the brain, due to increased central venous pressure (CVP). For example, transient increases in intrathoracic pressure due to coughing can cause spikes in the ICP.
- Intraabdominal pressure may be transmitted to the thorax (partially due to shifts in the diaphragmatic position). For example, one manifestation of abdominal compartment pressure is high peak airway pressures on the ventilator.
- Thus, elevation of either abdominal and/or thoracic pressure may threaten to increase the ICP.
- The most accessible measurement of intrathoracic pressure which is increasing ICP might be elevated airway pressures on the ventilator (e.g., elevated plateau pressure).
reduction of thoracic compartment pressures
- Depending on the etiology of the increased intrathoracic pressures, treatment may vary. Examples may include:
- (1) Reduction in airway pressures (e.g., reduction in PEEP as able – without causing derecruitment).(32440802)
- (2) Minimizing suctioning, as this may elicit coughing.
- (3) Drainage of large pleural effusions, if present.
- (4) Thoracic escharotomy in the context of severe burns.
reduction of abdominal compartment pressures
- Diagnosis and management of abdominal compartment syndrome is discussed further here. 📖
- If there is need for definitive control of compartment syndromes (e.g., severe abdominal compartment syndrome), abdominal fascial release could be the most accessible approach to this.
Follow us on iTunes
To keep this page small and fast, questions & discussion about this post can be found on another page here.
- Any time you are performing a lumbar puncture on a patient who might have elevated ICP, be sure to check an opening pressure.
- Avoid making major management changes based on transient fluctuations in the ICP.
- Don't forget to manage the cerebral perfusion pressure (CPP) along with the intracranial pressure.
- Don't become overly focused on a single aspect of ICP management (e.g., hypertonic saline) while forgetting that there are a half-dozen other ways to reduce the ICP.
- Please don't place a central access solely for the purpose of administering 3% saline.
Acknowledgement: Thanks to Dr. Richard Choi (@rkchoi) for thoughtful comments on this chapter.
Guide to emoji hyperlinks 
= Link to online calculator.
= Link to Medscape monograph about a drug.
= Link to IBCC section about a drug.
= Link to IBCC section covering that topic.
= Link to FOAMed site with related information.
= Link to supplemental media.
Review of seminal studies by The Bottom Line
- POLAR (2018) – Early prophylactic hypothermia did not benefit patients with traumatic brain injury.
- RESCUEicp (2016) – Decompressive craniectomy for patients with traumatic brain injury improved survival, while increasing the likelihood of poor neurological outcomes.
- BEST-TRIP (2015) – ICP monitoring in traumatic brain injury didn't improve outcomes, compared to clinical and CT scan monitoring.
- Eurotherm3235 (2015) – For patients with traumatic brain injury and elevated ICP, hypothermia increased a likelihood of poor neurological outcomes.
References
- 09115655 Kapadia FN, Jha AN. Simultaneous lumbar and intraventricular manometry to evaluate the role and safety of lumbar puncture in raised intracranial pressure following subarachnoid haemorrhage. Br J Neurosurg. 1996 Dec;10(6):585-7. doi: 10.1080/02688699646907 [PubMed]
- 17210899 Lenfeldt N, Koskinen LO, Bergenheim AT, Malm J, Eklund A. CSF pressure assessed by lumbar puncture agrees with intracranial pressure. Neurology. 2007 Jan 9;68(2):155-8. doi: 10.1212/01.wnl.0000250270.54587.71 [PubMed]
- 19565359 Castellani G, Zweifel C, Kim DJ, Carrera E, Radolovich DK, Smielewski P, Hutchinson PJ, Pickard JD, Czosnyka M. Plateau waves in head injured patients requiring neurocritical care. Neurocrit Care. 2009;11(2):143-50. doi: 10.1007/s12028-009-9235-7 [PubMed]
- 24411105 Lazaridis C. Plateau waves of intracranial pressure and mechanisms of brain hypoxia. J Crit Care. 2014 Apr;29(2):303-4. doi: 10.1016/j.jcrc.2013.12.002 [PubMed]
- 26438459 Stevens RD, Shoykhet M, Cadena R. Emergency Neurological Life Support: Intracranial Hypertension and Herniation. Neurocrit Care. 2015 Dec;23 Suppl 2(Suppl 2):S76-82. doi: 10.1007/s12028-015-0168-z [PubMed]
- 26704760 Lin AL, Avila EK. Neurologic Emergencies in the Patients With Cancer. J Intensive Care Med. 2017 Feb;32(2):99-115. doi: 10.1177/0885066615619582 [PubMed]
- 29329250 Kreitzer N, Huynh M, Foreman B. Blood Flow and Continuous EEG Changes during Symptomatic Plateau Waves. Brain Sci. 2018 Jan 12;8(1):14. doi: 10.3390/brainsci8010014 [PubMed]
- 30516597 Koenig MA. Cerebral Edema and Elevated Intracranial Pressure. Continuum (Minneap Minn). 2018 Dec;24(6):1588-1602. doi: 10.1212/CON.0000000000000665 [PubMed]
- 31659383 Hawryluk GWJ, Aguilera S, Buki A, et al. A management algorithm for patients with intracranial pressure monitoring: the Seattle International Severe Traumatic Brain Injury Consensus Conference (SIBICC). Intensive Care Med. 2019 Dec;45(12):1783-1794. doi: 10.1007/s00134-019-05805-9 [PubMed]
- 31773291 Changa AR, Czeisler BM, Lord AS. Management of Elevated Intracranial Pressure: a Review. Curr Neurol Neurosci Rep. 2019 Nov 26;19(12):99. doi: 10.1007/s11910-019-1010-3 [PubMed]
- 32227294 Cook AM, Morgan Jones G, Hawryluk GWJ, Mailloux P, McLaughlin D, Papangelou A, Samuel S, Tokumaru S, Venkatasubramanian C, Zacko C, Zimmermann LL, Hirsch K, Shutter L. Guidelines for the Acute Treatment of Cerebral Edema in Neurocritical Care Patients. Neurocrit Care. 2020 Jun;32(3):647-666. doi: 10.1007/s12028-020-00959-7 [PubMed]
- 32440802 Schizodimos T, Soulountsi V, Iasonidou C, Kapravelos N. An overview of management of intracranial hypertension in the intensive care unit. J Anesth. 2020 Oct;34(5):741-757. doi: 10.1007/s00540-020-02795-7 [PubMed]
- Nelson, S. E., & Nyquist, P. A. (2020). Neurointensive Care Unit: Clinical Practice and Organization (Current Clinical Neurology) (1st ed. 2020 ed.). Springer.
- 33896520 Tadevosyan A, Kornbluth J. Brain Herniation and Intracranial Hypertension. Neurol Clin. 2021 May;39(2):293-318. doi: 10.1016/j.ncl.2021.02.005 [PubMed]
- 34618757 Liotta EM. Management of Cerebral Edema, Brain Compression, and Intracranial Pressure. Continuum (Minneap Minn). 2021 Oct 1;27(5):1172-1200. doi: 10.1212/CON.0000000000000988 [PubMed]
- 34618760 Robinson CP. Moderate and Severe Traumatic Brain Injury. Continuum (Minneap Minn). 2021 Oct 1;27(5):1278-1300. doi: 10.1212/CON.0000000000001036 [PubMed]
- Jallo, J., I., & Urtecho, J. (2021). The Jefferson Manual for Neurocritical Care (1st ed.). Thieme.
- Louis ED, Mayer SA, Noble JM. (2021). Merritt’s Neurology (Fourteenth). LWW.
- 35058406 Rubiano AM, Figaji A, Hawryluk GW. Intracranial pressure management: moving beyond guidelines. Curr Opin Crit Care. 2022 Apr 1;28(2):101-110. doi: 10.1097/MCC.0000000000000920 [PubMed]
- 35816237 Hawryluk GWJ, Citerio G, Hutchinson P, Kolias A, Meyfroidt G, Robba C, Stocchetti N, Chesnut R. Intracranial pressure: current perspectives on physiology and monitoring. Intensive Care Med. 2022 Jul 11. doi: 10.1007/s00134-022-06786-y [PubMed]
- Albin, C. S. W., & Zafar, S. F. (2022). The Acute Neurology Survival Guide: A Practical Resource for Inpatient and ICU Neurology (1st ed. 2022 ed.). Springer.
- Naidech, A. M. (2022). Neurocritical Care (Cambridge Manuals in Neurology) (New ed.). Cambridge University Press.