CONTENTS
- Rapid Reference 🚀
- Diagnosis & symptoms
- Causes
- Evaluation
- Risk stratification
- Initial treatment (e.g., in ED)
- Subsequent therapeutic strategy
- Oops, the patient's sodium over-corrected
- Why not to use vaptans
- Podcast
- Questions & discussion
- Pitfalls
initial management package:
interpreting labs:

DDAVP clamp:

oral urea:

- Hyponatremia is a lab diagnosis.
- Consider repeating the lab before initiating therapy, especially if it doesn't match the clinical scenario or if other electrolytes are deranged. An aberrantly low sodium may result from drawing electrolytes upstream from a hypotonic infusion.
symptoms
- Severe:
- Seizure.
- Delirium, coma, herniation.
- Neurogenic pulmonary edema (figure below).
- Non-severe:
- Nausea, vomiting.
- Headache.
- Mild confusion.
- Dizziness, gait instability.
- Tremor, multifocal myoclonus.
- Hyperreflexia, muscle cramps.

pseudohyponatremia (serum osmolality is not actually low)
- Severe hyperglycemia.
- Triglyceride level >1,500 mg/dL.
- High protein level (multiple myeloma, IVIG).
- Exogenous osmoles:
- Contrast dye.
- Mannitol administration.
- Maltose (from IVIG).
- Sorbitol/glycine (used for surgical irrigation).
severe renal failure
- Typically occurs only when GFR <15 ml/min.
rare scenarios with water intake > solute intake
- Psychogenic polydipsia (especially in schizophrenia).
- Very rapid water intake (e.g., fraternity hazing, or water loading prior to a drug screen).
- Beer potomania (excessive beer intake with reduced solute intake).
- Elderly patients who eat a “tea-and-toast” diet, or anorexia (low solute intake).
hypovolemic hyponatremia
- Non-renal losses
- GI loss (vomiting, diarrhea, gastric tube drainage).
- Hemorrhage.
- Sweating, burns.
- Renal losses
- Diuretics (especially thiazides).
- Post-obstructive diuresis.
- Hypoaldosteronism or adrenal insufficiency (late).
- Cerebral salt wasting (possibly a form of hypoaldosteronism).
euvolemic hyponatremia
- Adrenal insufficiency (early).
- SIADH
- Medications (this list is incomplete; when in doubt evaluate the medication list using Medscape or Epocrates for possible causative agents).
- Vasopressin or DDAVP (these don't technically stimulate ADH release, but rather directly stimulate ADH receptors).
- NSAIDs.
- Chemotherapy (cyclophosphamide, vincristine).
- Psychiatric (antipsychotics, SSRIs, tricyclics, MAO inhibitors).
- Neurologic (carbamazepine, oxcarbazepine, valproate).
- Oxytocin, bromocriptine.
- Amiodarone
- Opioids
- Ecstasy (MDMA).
- Malignancy (especially small cell lung carcinoma).
- Neuropsychiatric disorder (virtually any).
- Severe respiratory failure.
- Pain or nausea (especially post-operative, or due to extreme exercise especially marathons).
- Medications (this list is incomplete; when in doubt evaluate the medication list using Medscape or Epocrates for possible causative agents).
- (⚠️ Hypothyroidism is often listed as a cause of hyponatremia, but this does not appear to be evidence-based.)(23902827)
edematous states (hypervolemic hyponatremia)
- Heart failure with low cardiac output.
- Cirrhosis.
- Nephrotic syndrome.
- Renal failure can also do this, if severe.
 labs to obtain for hyponatremia of unclear etiology
labs to obtain for hyponatremia of unclear etiology
- Full set of serum electrolytes (including Ca/Mg/Phos) & glucose.
- Serum osmolality. (Normal or increased level indicates pseudohyponatremia. This is an unusual finding, but is totally game-changing.)
- Cortisol level.
- Urine osmolarity & urine sodium.

interpretation of labs: a note of caution
- Traditional diagnostic algorithms often fail, because patients frequently have multifactorial hyponatremia (especially complex ICU patients). Therefore, patients have a tendency to break the rules and fall outside the boxes.
- Nonetheless, laboratory tests can often point us in the correct direction. Labs should always be combined with the history, medication evaluation, and physical examination.
urine osmolarity
- The preferred way to measure this is a direct measurement of urine osmolality. However, a crude estimate can be obtained by looking at the urine specific gravity. A specific gravity <1.010 suggests dilute urine, whereas a specific gravity >1.010 suggests concentrated urine.(21089176)
- Concentrated urine (urine osmolality >300 mOsm, specific gravity >1.010)
- Diagnostic significance
- Most hyponatremic patients will have a urine osmolality of >300 mOsm.
- This is nonspecific, revealing relatively little about the diagnosis.
- Therapeutic significance
- Concentrated urine indicates that the kidney is still retaining water. This indicates that the patient is not going to auto-correct their sodium.
- A very concentrated urine suggests that giving isotonic fluid could potentially worsen the hyponatremia (because the kidney may respond by retaining water and excreting sodium, a process termed “desalination”).
- Diagnostic significance
- Dilute urine (urine osmolality <<300 mOsm, specific gravity <1.010)
- Diagnostic significance:
- The kidney is secreting water – that's what it's supposed to do! The kidney is working correctly. This suggests one of the following situations:
- (1) Hyponatremia due to water intake >> solute intake (see purple differential above).
- (2) A patient in the recovery phase from any other cause of hyponatremia (e.g., the patient initially had hypovolemic hyponatremia, received volume resuscitation prior to urinalysis, and is currently auto-correcting their own sodium levels.)
- Therapeutic significance:
- Secretion of dilute urine will cause the patient's sodium to rise – so these patients will correct their own sodium levels.
- Production of large volumes of dilute urine is often a sign that the sodium is about to over-correct.
- Diagnostic significance:
urine sodium
- Low urine sodium (<20 mEq/L) suggests:
- Hypovolemic hyponatremia due to extra-renal volume loss.
- Hypervolemic hyponatemia (e.g. heart failure, cirrhosis).
- Intermediate urine sodium (~20-40 mEq/L)
- This represents a grey zone, provides no clear information.
- High urine sodium (>40 mEq/L) suggests:
- Euvolemic hyponatremia (SIADH or adrenal insufficiency).
- Hypovolemic hyponatremia due to renal salt wasting (e.g. diuretics, vomiting, or cerebral salt wasting).
This is not generally recommended (because it may make matters worse). However, it's very common for patients to get 1-2 liters of isotonic crystalloid shortly after presentation. If this happens, then the patient's response can be diagnostically illuminating:
- Sodium increase:
- This suggests that the patient has dilute urine (see above discussion).
- Sodium decreases:
- This proves the presence of concentrated urine (>>300 mOsm), as well as kidneys which are excreting salt. So essentially the patient holds onto the water and excretes the sodium (a process sometimes termed “desalination”).
- Most commonly, this implies the presence of SIADH.
Generally speaking, there are two things to worry about with a patient with hyponatremia. First, hyponatremia may worsen, leading to severe complications (e.g., seizure, cerebral edema, and herniation). Second, hyponatremia may overcorrect too rapidly, leading to osmotic demyelination.
risk for seizures/herniation due to hyponatremia 🤯
- (a) Symptoms of hyponatremia.
- (b) Acute hyponatremia.
- (c) Brain substrate:
- Premenopausal women (little extra space in cranium for brain to swell).
- Intracranial pathology (e.g. space-occupying lesion, cerebral edema).
- Known seizure disorder.
risk for osmotic demyelination syndrome due to rapid correction
- (a) Reversible cause of hyponatremia:
- Hypovolemia, adrenal insufficiency.
- SIADH due to a transient factor (e.g. pain, nausea, or medication).
- Situations where water intake > sodium intake (e.g. beer potomania, psychogenic polydipsia, tea-and-toast diets).
- (b) Chronicity & severity:
- Severe, asymptomatic hyponatremia is the most worrisome (especially Na <110 mM).
- Osmotic demyelination is rare if the initial sodium is >120-125 mM.(31601554)
- (c) Other risk factors for osmotic demyelination syndrome:(30181129)
- Hypokalemia (more on hypokalemic hyponatremia below).
- Cirrhosis.
- Alcoholism.
- Malnutrition.
- (d) High rate of sodium rise:
- >8-10 mM per day on average may cause osmotic demyelination.
- The greatest risk occurs if patients transition from chronic hyponatremia to hypernatremia. Hypernatremia should be aggressively avoided in patients who are recovering from hyponatremia.
⚠️ The malnourished, alcoholic patient will often have numerous risk factors for central pontine myelinolysis (hypokalemia, cirrhosis, alcoholism, and malnutrition).
Initial management is actually pretty simple:
indications for a bolus of IV hypertonic therapy:
- The strongest indication is symptomatic hyponatremia, e.g.:
- Seizure
- Confusion
- Headache, nausea, vomiting
- Dizziness, gait instability, tremor, multifocal myoclonus
- Hypertonic therapy may also be considered for patients with profound hyponatremia, in whom any further worsening could be life-threatening.
how to give a bolus of hypertonic fluid:

- (option #1) Hypertonic bicarbonate (more on this here)
- Hypertonic bicarbonate is defined here as 1 mEq/ml sodium bicarbonate, which is generally found in 50-ml ampules on crash carts. This has the advantage that it's generally the fastest medication to obtain in an emergency.
- 1 mEq/ml sodium bicarbonate has the same tonicity as would 6% NaCl.
- A typical dose is two ampules (100ml) of hypertonic bicarbonate (equivalent to giving ~200 ml of 3% saline, which will raise the serum sodium by ~3 mM). For smaller patients, 1 or 1.5 ampules (50-75 ml) might be more appropriate (depending also on the clinical context and the urgency of increasing the sodium).
- Ampules of bicarbonate should generally be infused slowly (e.g. each one over 5 minutes). Bicarbonate is contraindicated in patients with metabolic alkalosis, which would be exacerbated by the bicarbonate.
- (option #2) Hypertonic saline
- 3% saline may be provided in a dose of 2 ml/kg body weight (e.g., ~150 ml).
- This is the traditional therapy for hyponatremia.
- The main drawback of 3% saline is that it often takes a long time to receive from the pharmacy.
- Please note that 3% saline is safe to administer through a peripheral line. 3% saline does NOT require placement of a central line (30745195, 29472509, 28372499, 27965228).
follow-up repeat electrolytes
- Repeat a full electrolyte panel after administration of hypertonic therapy.
- The goal is to increase the Na by about 3-5 mM, which should cause clinical improvement.
- If symptoms persist:
- If the sodium has increased by <4 mM, then an additional round of hypertonic therapy may be needed (with a goal of increasing the sodium by ~4-6 mM total).
- If the sodium has increased by 6 mM and the symptoms have not resolved, then hyponatremia isn't the cause of the patient's symptoms. Evaluate further for alternative or additional problems.
target rates of sodium correction
- Target increasing the sodium by 6 mEq/L every 24 hours (and no more than 8 mEq/L). Note that if the sodium was increased by 4-6 mM acutely, this is included within the 6 mM increase which is desired over the first 24 hours.
- For example: If you raise the sodium by 6 mM to treat symptomatic hyponatremia, the goal over the next 24 hours is to maintain the sodium at that same level without any further rise.
- Excessively rapid correction runs a risk of osmotic demyelination. This is extremely debilitating and won't be obvious immediately – hence the necessity of controlling the rise in sodium.
- The key is the net rate of correction. If the sodium corrects too rapidly but then you rapidly lower the sodium back to it's target value, that's OK.
- For patients with acute hyponatremia (definitely developing over <<48 hours), faster rates of sodium correction may be safe. For example acute hyponatremia occuring during a marathon or following MDMA intoxication can be rapidly corrected. However, in nearly all patients presenting to the hospital with hyponatremia, this should be assumed to be chronic and treated accordingly.
the physiology of sodium overcorrection
- Over-correcting the sodium is almost never due to the practitioner's giving too much sodium chloride.
- Common causes of over-correction:
- 1) The underlying cause of hyponatremia is reversed. This causes the body to suddenly excrete a lot of free water, thereby causing the sodium to rise. This form of auto-correction is by far the most common cause of over-correction. The DDAVP clamp may be used to avoid this pitfall (next section).
- 2) KCl administration, without taking into account the effect this will have on sodium (more on this below).
- 3) Treatment of hyponatremia with vaptans (more on this below).
- Note, also, that over-correction is easily treated if it occurs (more on this below). Patients who overcorrect will do fine if their sodium is immediately lowered. The patients who develop osmotic demyelination are patients who over-correct and nothing is done about it.
use caution with predictive equations
- Equations can be used to predict the effect of a fluid on the patient's sodium level (e.g. MDCalc here).
- These equations treat the body as a passive receptacle which receives IV fluid and does nothing with it. This assumption would work well for patients in anuric renal failure.
- Predictive equations will fail most spectacularly for patients with reversible hyponatremia. In this scenario, patients may rapidly excrete lots of dilute urine and thereby over-correct their sodium spontaneously (much faster than would be predicted).
- In contrast, predictive equations tend to work much better for patients on the DDAVP clamp, because these patients won't produce much urine.
- Bottom line on predictive equations:
- These are a good starting place for estimating how much fluid to give in various situations. So we do recommend using them (especially in conjunction with the DDAVP clamp).
- Predictive equations don't work perfectly – so you still need to monitor the patient's electrolytes to ensure that therapies are working.
general concept
- This section discusses management of situations where the main concern is overcorrection of the sodium, leading to osmotic demyelination.
- Risk factors for sodium overcorrection & osmotic demyelination are listed above.
- The DDAVP clamp technique can be useful in patients at high risk for over-correction, or in patients who have already started to over-correct.
general concept of the DDAVP clamp
- Administration of DDAVP prevents the kidneys from secreting free water. This takes the patient's kidneys out of the equation, preventing the patient from auto-correcting. The patient essentially becomes like a passive container, with very predictable behavior:
- Administration of hypotonic fluid (e.g. water, juice) will cause the sodium to decrease.
- Administration of hypertonic fluids (e.g. 3% saline, potassium tablets) will cause the sodium to increase.
- If there are no inputs, the sodium should stay roughly stable.
how to perform the DDAVP clamp

- Give DDAVP 2 micrograms IV q8 hours scheduled.
- The patient's fluid intake must be strictly controlled.
- With DDAVP, if the patient drinks too much they will lower their sodium.
- To begin with, restrict any fluid intake (dry foods are OK). After 6-12 hours you may liberalize a bit.
- Note that the patient's sense of thirst is due to hyponatremia, so there is no safe way to get rid of this immediately.
- Calculate the rate of 3% saline required to achieve your target sodium.
- Figure out how much more you want the sodium to increase over the next 24 hours.
- The effect of 3% sodium on your patient's sodium can be calculated using this MDCalc equation.
- Infuse 3% saline via a peripheral vein (you don't need a central line; 3% saline is safe for peripheral administration).
- Follow electrolytes every 4-6 hours to ensure that the sodium is on track.
- If the sodium is too low, increase the rate of 3% saline.
- If the sodium is too high, calculate the amount of free water required to decrease it back to target using this formula. This volume of free water may be given intravenously (as D5W) or orally (if the patient is able to drink).
- This is tedious, but effective.
removing the DDAVP clamp
- Once the sodium is close to normal (e.g. >125 mM), the DDAVP clamp may be discontinued. About 10 hours later, the patient's diet may be gradually liberalized.
- One of two things will happen at this point:
- (a) If the cause of hyponatremia has been eliminated (e.g. hypovolemia), then the sodium will rapidly increase to normal and stay within a normal range. You're all set.
- (b) If the patient still has a cause for hyponatremia (e.g. chronic SIADH), their sodium may start falling again. At this point, they would require maintenance therapy for SIADH (e.g. furosemide and oral salt tabs, or oral urea).
More information on the DDAVP clamp is located here.
The DDAVP clamp-bolus strategy is a slight modification of the DDAVP clamp (section above). In the DDAVP clamp-bolus strategy, sodium is adjusted using discrete boluses of fluid, rather than a continuous infusion:
Advantages of the DDAVP clamp-bolus strategy (as compared to a strategy of DDAVP clamp plus a continuous infusion of 3% saline):
- Patients can rapidly and definitively be corrected back to their goal trajectory.
- Changes in therapy will take effect prior to the next sodium check, creating a tight feedback loop that allows for precise management in real time (as opposed to changing the rate of an infusion of 3% saline, which won't take effect for 5-10 hours).
The volume of boluses may be calculated as follows:
- 3% saline: The Madias equation can be used, with this calculator from MDCalc.🧮 The calculator can be set to determine the volume of 3% saline to increase the sodium by 1 mM; this may will need to be scaled up to match the desired increase in sodium. With the exception of initial treatment for symptomatic hyponatremia, it's probably inadvisable to use boluses of 3% saline that are larger than 250 ml.
- Hypertonic bicarbonate (1 mEq/ml): This has the same tonicity as 6% NaCl. Start by calculating the volume of 3% saline that would be required, as described above. Then divide by two to obtain the equivalent volume of hypertonic bicarbonate.
- D5W: The volume of water may be calculated using the free water deficit (setting the desired sodium level as the patient's goal sodium at that point in time).🧮
This section describes treatment of SIADH with an ongoing cause which is difficult or impossible to remove (e.g. malignancy, severe brain injury, or medications). Always be sure to look for all potential reversible causes of SIADH (e.g., medications listed above, nausea).
There are two preferred treatment strategies for chronic SIADH: oral urea or loop diuretic plus sodium. In either case, a moderate amount of fluid restriction should also be employed.
oral urea (aquaresis)

- Oral urea is emerging as a front-line therapy for SIADH.
- The kidneys will excrete any exogenous urea in the urine, along with water. Thus, oral urea intake functions as an aquaretic that forces the net elimination of water from the body. The volume of water removed is proportional to the amount of urea administered, leading to a finite and controlled removal of water (equation above). Urea is generally cleared from the body within ~12 hours.
- Oral urea has been used for SIADH for decades in Europe, and has long been recommended by the European guidelines on hyponatremia. Recently, several studies have confirmed that oral urea is a safe and effective therapy for SIADH (22378162, 30181129, 30614552, 30868608).
- Contraindications to urea are as follows:
- Inability to tolerate PO intake.
- Cirrhosis with hepatic encephalopathy.
- Severe renal failure.
- Dosing and monitoring is outlined in the figure below.(further discussion of aquaresis here)
- The starting dose is 15-30 grams (with a dosing range of 7.5-90 grams/day).

traditional therapy: loop diuretic + sodium
- This is an effective strategy for SIADH. However, it's generally not preferred compared to oral urea for the following reasons:
- Combination therapy is trickier (if the doses of loop diuretic and sodium aren't balanced correctly, this may provoke volume overload or volume depletion).
- Frequent doses of loop diuretic may cause frequent urination at night, interfering with sleep.
- Nonetheless, this treatment may be useful in situations where oral urea is contraindicated, or unavailable.
- General concept:
- (i) These patients are usually in sodium balance, so they shouldn't retain sodium. As sodium is excreted, it will pull water out of the body along with it. The amount of water loss is equal to the osmotic load of the sodium divided by the urine osmolarity.
- (ii) Continuous exposure to a loop diuretic will wash out the concentration gradient in the kidney, causing the urine osmolarity to decrease. Thus, even though the kidney is “trying” to retain water, it's less able to achieve that. Washing out the kidney concentration gradient will increase the amount of water which is pulled out of the body due to sodium administration (in part #i).
- Nuts & bolts:
- (1) Loop diuretic (e.g. furosemide) should be given frequently enough so that the kidney doesn't escape in between doses (e.g. 20 mg IV furosemide q6hr). As patients stabilize, this may be weaned down or off (furosemide will augment the efficacy of the NaCl, but it's not mandatory for it to work).
- (2) Sodium intake is increased. In acutely ill patients, this is usually achieved with a 3% NaCl infusion. As chronic maintenance therapy or in less emergent situations, oral salt tabs may also be used (a typical dose would be 3 grams TID with meals).
- (3) Monitor fluid balance, to make sure that the patient doesn't become volume overloaded or depleted.
general comments
- Heart failure rarely causes profound hyponatremia. Rather, there is usually a fairly stable sodium of ~120-135 mM.
- Hyponatremia due to heart failure is usually chronic and is not a life-threatening process.
- Exactly when treatment is beneficial is unclear. The best approach to chronic, asymptomatic hyponatremia is often to provide no specific therapy for the hyponatremia. Instead, the primary focus of therapy should remain the heart failure.
- There is a risk of focusing too much energy on treating the hyponatremia, and not enough energy on treating the underlying heart failure. For example: It's counter-productive for a cardiology service to refuse admission of a heart failure patient solely due to moderate hyponatremia.
physiology
- Inadequate cerebral perfusion stimulates the brain to produce antidiuretic hormone (ADH), leading to water retention.
- Thus, hyponatremia in heart failure is a reflection of poor systemic perfusion. This may explain why hyponatremia is a prognostic sign of poor long-term outcome.
treatments
- This depends on a clinical assessment of the patient's perfusion and volume status. For example:
- (1) Sometimes acute hyponatremia may be a feature of cardiogenic shock (deteriorating systemic perfusion).
- Treatment here will depend on systemic hemodynamic assessment.
- Sometimes hypoperfusion may be due to volume depletion – such patients may benefit from judicious volume repletion.
- Some hypoperfused patients could benefit from therapies such as inotropes (or withdrawal of beta-blockers). For more on this, see the chapter on cardiogenic shock.
- (2) Chronic hyponatremia is often associated with volume overload.
- For patients with marked volume overload, furosemide is an excellent therapy. Furosemide stimulates the production of dilute urine, causing loss of both water and sodium (but generally more water than sodium). This will improve the volume status and also increase the sodium concentration.
- For patients with mild volume overload and substantial hyponatremia, oral urea could be a consideration. Urea will cause a pure water loss (aquaresis), so it will increase the sodium level with less volume loss. A figure describing how to use oral urea is shown above.
- For patients with hyponatremia and severe refractory congestion that doesn't respond to conventional doses of loop diuretics, a combination of simultaneous loop diuretic and hypertonic saline may be trialed (i.e. hyperdiuresis, typically achieved by administering 150 ml 3% saline plus 250 mg furosemide IV every 12 hours).
general comments
- Cirrhosis rarely causes severe hyponatremia. Similar to heart failure, there is usually a fairly stable sodium of ~120-135 mM.
- This is usually chronic and not a life-threatening process.
- Exactly when treatment is beneficial is unclear. The best approach to chronic, asymptomatic hyponatremia could be to do nothing.
treatment
- As in heart failure, careful assessment of perfusion and volume status should be performed.
- Rare patients may be hypovolemic, in which case judicious volume resuscitation may be considered.
- Furosemide diuresis is often a good option for patients with volume overload. Furosemide stimulates the production of dilute urine, causing loss of both water and sodium (but generally more water than sodium). This should improve the volume status and also increase the sodium concentration.
- Oral lactulose is a good option for patients with any degree of hepatic encephalopathy. Lactulose causes wasting of water via the gut (which will improve the hyponatremia), while simultaneously treating hepatic encephalopathy (more on this here).
- Oral urea might be considered in patients without any history of hepatic encephalopathy, but its use in cirrhosis is controversial. There is concern that urea could be converted into ammonia by gut bacteria, promoting the development of hepatic encephalopathy.
- Overall, urea is probably a 2nd or 3rd line agent here (and potentially inferior to lactulose). If urea is used, it should be closely monitored.
potassium increases tonicity as much as sodium does
- Administration of KCl will increase the sodium concentration just as much as NaCl would (they have the same impact on tonicity).
- This is a bit counterintuitive, because the potassium levels don't increase much. However, potassium entry into cells is paired with the exit of sodium, such that the sodium levels will rise.
- 50 mEq of oral KCl will have about the same effect as 100 ml of 3% NaCl.
- The effect of intravenous forms of potassium will depend on exactly how they are formulated.
estimating the effect of oral KCl supplementation on serum sodium

- The impact of oral potassium tablets on serum sodium can be estimated as above (if we approximate the total body water as roughly equal to 55% of the patient's weight).
- This effect can be more than expected, for example:
- In a very small 40-kg patient, an oral dose of 40 mEq KCl could increase the sodium by ~2 mEq/L.
- The potassium deficit increases exponentially as potassium levels fall (explored further here). Thus, a 70-kg patient with a potassium of 2.5 mM could have a potassium deficit of ~500 mEq. Administration of this quantity of KCl orally would increase sodium by ~12 mM!
clinical significance
- Oral repletion of KCl will have a greater impact on serum sodium than we generally recognize. This should be accounted for in patients with significant hypokalemia.
- Failure to consider the osmotic effect of KCl is one driver of unexpected over-correction of hyponatremia.
- In some patients with moderate hyponatremia and moderate hypokalemia, oral repletion of KCl will simultaneously address both problems.
Over-correction will occasionally happen. With proper management, this can be rapidly fixed and patients will do fine.
key overcorrection concept #1 = delayed deterioration
- Correction at rates >8-10 mM per day may cause osmotic demyelination syndrome (previously termed central pontine demyelination). This is a permanent, debilitating condition which must be avoided.
- Osmotic demyelination doesn't occur immediately. So, if the patient over-corrects and still has a normal neurologic examination, this doesn't mean that they couldn't potentially develop osmotic demyelination soon.
- Even if the over-correcting patient looks OK, they're still in trouble – and this still needs to be actively managed.
key overcorrection concept #2 = you need to get back on track
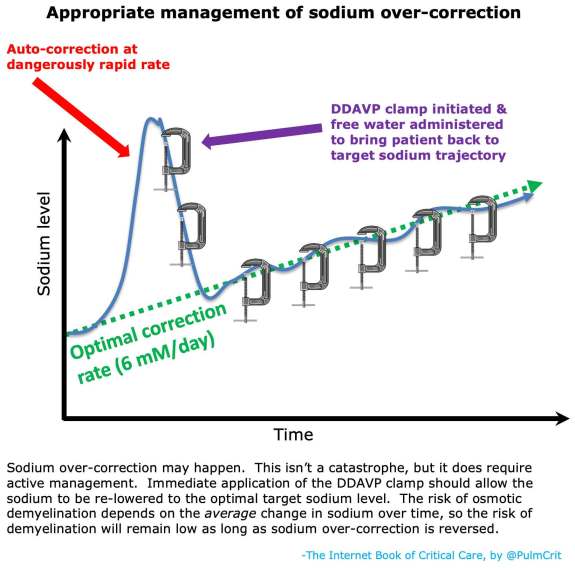
- What matters is the average rate of sodium rise over time. So if the sodium skyrockets up, that's OK – as long as you subsequently bring it back to target.
- The best approach is immediate initiation of the DDAVP clamp:
- Start DDAVP 2 micrograms IV q8hr
- Calculate the amount of free water needed to bring the patient back down to their target sodium level (you can use this MDCalc formula for free water deficit). This should be given fairly quickly, with a goal of bringing the patient back to their target sodium as soon as possible.
- Follow the sodium closely. Use either water or hypertonic sodium to adjust the sodium to your target trajectory.
- More about the DDAVP clamp above and also here.

Historically, folks have often been afraid of using 3% saline (yet felt more comfortable using vaptans). This is completely backwards. 3% saline is actually extremely safe. Meanwhile, vaptans have a unique capacity to cause runaway water loss and overcorrection. This isn't just my opinion – the European guidelines for hyponatremia recommend against vaptan use for treatment of hyponatremia.
vaptans induce a state of uncontrolled diabetes insipidus
- Vaptans block aquaporin water channels in the kidneys, causing uncontrolled excretion of water by the kidneys. This will mimic nephrogenic diabetes insipidus. Giving a vaptan is thus the opposite strategy compared to the DDAVP clamp.
- There are several problems with this:
- 1) Water loss is uncontrolled and unpredictable. Patients can correct their sodium much faster than would be desired.
- 2) Patients may develop overshoot hypernatremia. This is particularly dangerous regarding the risk of osmotic demyelination.
- Vaptans are also extremely expensive and some may be potentially hepatotoxic. But these problems pale in comparison to the simple truth that the mechanism of action of vaptans is inherently dangerous and uncontrolled.
Follow us on iTunes
The Podcast Episode
Want to Download the Episode?
Right Click Here and Choose Save-As
To keep this page small and fast, questions & discussion about this post can be found on another page here.

- Don't forget to implement free water restriction for all hyponatremic patients (at least early on, while the dust is settling).
- Don't use vaptans. It's that simple, just don't use them.
- If you use the DDAVP clamp, be sure to restrict the patient's fluid intake. The combination of DDAVP plus unrestricted fluid intake can worsen the patient's hyponatremia.
- Patients with symptomatic hyponatremia should be treated with hypertonic fluid (either 3% saline or hypertonic sodium bicarbonate). Don't use normal saline here, because if the patient has SIADH this may exacerbate their hyponatremia.
- If the patient's sodium rises too quickly, don't give up (“well the patient's ok, they seem to have tolerated it”). Patients may look OK for a while, but later develop osmotic demyelination. If the patient's sodium over-corrects, give them DDAVP and water, and reduce the sodium to the appropriate target.
Guide to emoji hyperlinks 
= Link to online calculator.
= Link to Medscape monograph about a drug.
= Link to IBCC section about a drug.
= Link to IBCC section covering that topic.
= Link to FOAMed site with related information.
= Link to supplemental media.
References
- 21089176 Imran S, Eva G, Christopher S, Flynn E, Henner D. Is specific gravity a good estimate of urine osmolality? J Clin Lab Anal. 2010;24(6):426-30. doi: 10.1002/jcla.20424 [PubMed]
- 22378162 Coussement J, Danguy C, Zouaoui-Boudjeltia K, Defrance P, Bankir L, Biston P, Piagnerelli M. Treatment of the syndrome of inappropriate secretion of antidiuretic hormone with urea in critically ill patients. Am J Nephrol. 2012;35(3):265-70. doi: 10.1159/000336716 [PubMed]
- 24569125 Spasovski G, Vanholder R, Allolio B, Annane D, Ball S, Bichet D, Decaux G, Fenske W, Hoorn EJ, Ichai C, Joannidis M, Soupart A, Zietse R, Haller M, van der Veer S, Van Biesen W, Nagler E; Hyponatraemia Guideline Development Group. Clinical practice guideline on diagnosis and treatment of hyponatraemia. Eur J Endocrinol. 2014 Feb 25;170(3):G1-47. doi: 10.1530/EJE-13-1020 [PubMed]
- 30181129 Rondon-Berrios H, Tandukar S, Mor MK, Ray EC, Bender FH, Kleyman TR, Weisbord SD. Urea for the Treatment of Hyponatremia. Clin J Am Soc Nephrol. 2018 Nov 7;13(11):1627-1632. doi: 10.2215/CJN.04020318 [PubMed]
- 30596344 Peri A. Management of hyponatremia: causes, clinical aspects, differential diagnosis and treatment. Expert Rev Endocrinol Metab. 2019 Jan;14(1):13-21. doi: 10.1080/17446651.2019.1556095 [PubMed]
- 30614552 Lockett J, Berkman KE, Dimeski G, Russell AW, Inder WJ. Urea treatment in fluid restriction-refractory hyponatraemia. Clin Endocrinol (Oxf). 2019 Apr;90(4):630-636. doi: 10.1111/cen.13930 [PubMed]
- 30868608 Nervo A, D'Angelo V, Rosso D, Castellana E, Cattel F, Arvat E, Grossi E. Urea in cancer patients with chronic SIAD-induced hyponatremia: Old drug, new evidence. Clin Endocrinol (Oxf). 2019 Jun;90(6):842-848. doi: 10.1111/cen.13966 [PubMed]
- 31232710 Hoorn EJ, Spasovski G. Recent developments in the management of acute and chronic hyponatremia. Curr Opin Nephrol Hypertens. 2019 Sep;28(5):424-432. doi: 10.1097/MNH.0000000000000528 [PubMed]
- 31601554 Rondon-Berrios H. Therapeutic Relowering of Plasma Sodium after Overly Rapid Correction of Hyponatremia: What Is the Evidence? Clin J Am Soc Nephrol. 2020 Feb 7;15(2):282-284. doi: 10.2215/CJN.04880419 [PubMed]





