
By Katrina Augustin, MD, BSN
Peer Reviewed by Eric Leiendecker, MD; Divya Gupta, MD; and Lewis McLean, MD
Drawings by Tammy Lin, MD
So LVADs…
You know that patient that you NEVER want to see show up in your emergency department—the one that makes you tachycardic…


AND…. the crashing LVAD patient.
Due to the improving LVAD design, decrease in adverse effects, and increased indications for LVAD implantation—patients with these devices are increasing in prevalence. Hence, it is imperative that emergency providers can not only troubleshoot these devices but also treat emergent pathology that arises.

The combination of a lack of suitable organs for transplant, in addition to the increasing number of patients not eligible for transplant (advanced age/comorbidities), has contributed to the increased demand for durable support with an LVAD. The results of the REMATCH trial demonstrating improved quality of life as well as survival benefit with LVAD when compared to medical therapy further lend support to this therapy. The HeartMate 3 (HM3) is implanted as bridge to transplantation (BTT), bridge to decision (or candidacy), as well as destination therapy (DT). [PMID: 31692113]
This post is meant to provide a basic review and update on durable mechanical LVAD support with a focus on HeartMate 3 (HM3).
This post will cover:
- HM3 introduction
- Obtaining vital signs
- LVAD assessment
- LVAD related complications + disease processes requiring special treatment in this population
- Management of the LVAD patient in extremis (only have 5 minutes—read this)
HM3 Introduction
HM3 is the most common durable LVAD currently being manufactured and implanted in the United States. While there are still some axial-flow HeartMate 2 and centrifugal-flow
HeartWare LVADs in circulation, recent studies showed improved outcomes with HM3 when compared to these devices prompting a transition to HM3. [PMID: 34980564]

The MOMENTUM 3 trial, which randomized patients to the axial-flow HM2 LVAD versus the intrapericardial centrifugal-flow HM3 LVAD, found that patients with HM3 were more likely to achieve the primary endpoint of survival to 2 years free of a disabling stroke or need for a reoperation/replacement of a malfunctioning device when compared to patients with HM2 (76.9% vs 64.8, p<0.001). Furthermore, they also found a lower incidence of GI bleeding with HM3.
A retrospective analysis by Mueller et.al comparing HM3 with the other centrifugal pump on the market, the HeartWare HVAD, found an increased risk of cerebral bleeding in the HeartWare cohort. Increased adverse events lead to the FDA issuing a recall with no further production or implantation of HeartWare HVADs.

The improved outcomes with HM3 are attributed to modifications in pump design leading to an increase in hemocompatibility with DECREASED activation and/or destruction of blood elements.

Okay great so the HM3 LVAD is better and there are more patients receiving them, but what I really care about is what do I do with them when they show up in my emergency department?
Step 1: Obtain Vital Signs

Now on to vitals—with a continuous flow LVAD, patients may not have a palpable pulse unless there is enough native cardiac function to generate pulsatile flow thus making standard vital sign measurement difficult.
Obtain the mean arterial pressure (MAP) equivalent – known as return to flow (RTF)
-
- Place pencil doppler probe over the brachial artery (or ultrasound with doppler)
- Inflate BP cuff to >30 mmHg over where doppler flow last heard or visualized (if using CFD on US)
- Deflate cuff till flow heard or seen again which is the “return to flow”
- Some patients may have enough native cardiac function to generate pulsatile flow with resultant BP able to be measured by automatic BP monitors. The HM3 artificial pulse (speed adjustment every 2 seconds) may contribute to some pulsatility.
- Low threshold to place arterial line if difficulty obtaining MAP—waveform may be flatline with only variations from HM3 artificial pulse

- Heart rate (HR):
- As patient may not have palpable pulse, easiest to place on monitor to assess rhythm and rate simultaneously
- Majority will have and ICD +/- pacemaker
- Pulse oximetry:
- Potentially inaccurate due to lack of pulsatile flow, although may pick up pulsatility from artificial pulse
- If in extremis, may need to obtain ABG from arterial line
- End Tidal CO2:
- Useful in intubated patients in extremis to give insight into low flow state such as severe shock and cardiac arrest where end tidal can reflect systemic perfusion
- If the ETCO2 is <20 and MAP <50 or undetectable – Start CPR/ACLS (per the 2017 AHA ACLS update for LVAD) PMID: 28533303
Step 2: Contact LVAD Coordinator/Nearest LVAD Center Early!
LVAD patients will typically have an emergency contact number to reach their LVAD coordinator which is commonly a nurse or NP depending on the institution. The coordinator can assist with identification of LVAD alarms, controller exchanges, as well as mobilizing appropriate resources (LVAD engineer) and connecting you with the appropriate specialist. If no emergency contact number is available, contact the nearest LVAD center. It is imperative to call early to mobilize the appropriate resources especially in critically ill patients.
Step 3: Assess the LVAD
Yes, you DO need to ACTUALLY evaluate the LVAD. 😂
Always ensure that the driveline is intact and connected to the controller, the controller is not alarming, and that the LVAD is connected to a power source (batteries, mobile power unit module). See guide section on HM3 for directions on battery exchanges and alarms.

Then you are ready to assess the LVAD parameters. HM3 will report four parameters on the controller that are important as they can provide insight into what is going on with the patient.
But first, while it is helpful to know normal parameter ranges, it is most important to determine the patient’s “normal parameters” as any deviation can signify badness.
Speed
in revolutions per minute (RPM), is the only parameter that is set.
Power
is a direct measure of pump current and voltage. It indicates the “work” being done by the pump. Changes in speed, flow, and physiological demand can alter the power. A buildup of clot on the rotor or a high flow condition such as aortic regurgitation increase power, whereas an obstruction to flow (inflow cannula obstruction) will lead to decreased power.
Flow
which can be thought of as the “LVAD cardiac output” is an estimated value that is calculated from power, RPM (speed), and the patient’s hematocrit. Flow is directly proportional to speed and inversely proportional to the LVAD inflow to outflow pressure gradient.
Simplified: an INCREASE in RPM will cause and INCREASE in FLOW. While an INCREASE in the pressure gradient between the LV (inflow) and aorta (outflow) will cause a DECREASED flow.
Okay easy enough right?
But wait, the pressure gradient between the LV and aorta is not constant. During diastole the left ventricular pressure (LVP) and the aortic pressure (AoP) are significantly different leading to and INCREASED pressure gradient and thus DECREASED flow, conversely during systole the LVP and AoP are more similar leading to DECREASED pressure gradient and hence INCREASED flow.
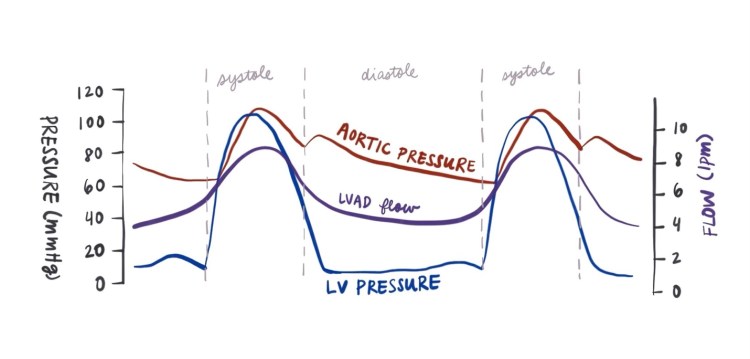
So, while there is continuous flow through the device, the flow will be variable. This variation in flow can lead to some pulsatility and is described by the pulsatility index (PI).
In most clinical scenarios, the PI can be used to represent the native contractility of the heart with increased contractility causing larger fluctuations in flow and thus increased PI. The average flow is assessed over 15 seconds. A change of >45% from baseline flow will trigger a PI event (with transient drop in speed) and is often due to transient changes in preload or afterload due to movement, coughing, arrythmias, etc. Most PI events will NOT trigger suction events (collapse of the LV normally due to low LV preload with hemodynamic instability) but can if they are sustained.
So that was a lot of talk about changes in pressure and flow, why do I as a clinician care?
Clinical changes such as decreased preload (hypovolemia), decrease LV pressure thus increasing the LV to aorta pressure gradient with resultant decreased flow and vice versa. So, giving a fluid bolus to a hypovolemic patient will increase LV pressure and increase flows. Hence, we say the LVAD is preload dependent. Similarly, an increased afterload (hypertension), causes an increased LV to aorta pressure gradient with resultant decreased flow and we say the LVAD is afterload sensitive.
And because it is so important—said another way…
*Flow through an LVAD is dependent on its specific HQ curves (head pressure (H) vs flow (Q)) and speed (RPM). Slope of the HQ curve dictates variations in flow for each pressure gradient. HM3 is unique to other LVADs in that its HQ curve has a flat slope at low flows leading to large variations in flow for small changes in pressure and at higher flows (>4L) has a steeper slope where there is less variation in flow for a set pressure change. HM3’s HQ curve is unique because it measures power of the rotor directly and thus can estimate flow much more accurately at low flows. These variations in flow alter the PI and thus explain why PI can be variable depending on flows in conditions like hypovolemia, RV failure (with underfilling of LV). PMID: 34980564
Step 4: Identify& Treat LVAD-related Complications & Disease Processes Requiring Special Treatment in this Population
Despite the improvement in pump design, patients with a HM3 LVAD are still prone to many complications. It is imperative that first line providers can identify and treat these complications as well as other disease conditions requiring special treatment in this population. [PMID: 28559233, PMID: 30202496]
Suction Events
- Collapse of the LV with interventricular septal deviation to the left due to negative pressure from continuous flow LVAD
- Risk factors: anything that decreases LV preload, malpositioned inflow cannula directed towards septum
- Suction events can cause arrhythmias, low LVAD output, and hemodynamic instability. HM3 will automatically decrease speed transiently in an attempt to resolve suction event.
- ALWAYS perform bedside cardiac POCUS if concerned for a LVAD suction event as treatment is very different depending on the etiology
GI Bleeding (GIB)
- While the MOMENTUM 3 trial showed a decreased risk of bleeding in HM3 compared to HM2 this is still a common complication with incidence ranging from 9.7% (ELEVATE) to 24.5% (MOMENTUM 3) in trials.
Special Considerations for GIB in setting of LVAD
-
- Consider endoscopy/colonoscopy for AVM identification/treatment
- Avoid reflexive blood transfusions if patient is hemodynamically stable, especially if patient is bridge to transplant, as can increase risk of sensitization making transplant more difficult
- Collaborate with LVAD team when considering AC reversal to allow them to guide treatment
- Large volume resuscitation, such as MTP, can precipitate RV failure so proceed with caution—low threshold to perform POCUS for RV function/volume status
Stroke
- Reduced risk with HM3 compared to other LVADs however still approximately 10% incidence by 2 years
- Increased risk with hypertension (RTF/MAP>90) and supratherapeutic INR (hemorrhagic CVA)
Special Considerations for stroke management in setting of LVAD
-
- Stat CT head & CTA head/neck +/- CT perfusion to assess for large vessel occlusion (LVO) amendable to mechanical thrombectomy
- HM3 LVAD is NOT MRI compatible
- Ischemic strokes: Majority of patients have contraindications to systemic TPA including warfarin therapy with INR>2
- Hemorrhagic strokes: Early NSGY/stroke team/LVAD team consultation important to determine coagulation reversal and other treatments
Pump Thrombosis
- Associated with significant morbidity and mortality. Device exchange typically needed.
- Prevalence significantly reduced with HM3 compared to other LVADs (only 1.5% at 2 years)
Special Considerations for pump thrombosis
-
- Suspect with elevated power (will also have paradoxically elevated flows since calculated from power) in the setting of hemolysis +/-low output syndrome
- Assess for evidence of hemolysis: elevated LDH, low haptoglobin, elevated plasma free hemoglobin, hemoglobinuria, and acute kidney injury
- Consult cardiothoracic surgery for management: AC/pump exchange/transplant or possibly TPA
RV Failure
- Despite advances in pump design with HM3, RV failure continues to be the “Achilles heel” of LVAD support with an incidence of 9-40%. PMID: 34980564
Special Considerations for RV failure in LVAD patients
-
- Optimize preload: consider careful diuresis, avoid fluids unless clear history of volume depletion and evidence on exam
- Support RTF (MAP) to maintain RV perfusion if hypotensive (epinephrine/vasopressin/norepinephrine)
- Decrease RV afterload: prevent hypoxia/hypercarbia, consider inhaled pulmonary dilators
- Inotropes if impaired RV contractility (low dose epinephrine, dobutamine, milrinone)
- Avoid positive pressure ventilation/intubation: can precipitate worsening RV failure
- Consult: May require mechanical circulatory support, including VA ECMO as bridge to RVAD support PMID: 33717597
Infection
- 20% of patients develop a LVAD related infection in the 1st Staphylococcus most common, however pseudomonas and other GNRs are also frequent pathogens
- Trauma/traction on driveline disrupting seal between skin/driveline allow portal of entry. Can also develop pump pocket/pump infection or endocarditis.
- Special Considerations for LVAD related infections
- Always consider LVAD related infections as well as typical infections when these patients present with sepsis. Use caution with fluid resuscitation to avoid precipitating RV failure with rapid administration of large volumes of crystalloid
- Obtain driveline culture as well as blood cultures prior to antibiotic administration
- Consider imaging to assess for infectious foci around driveline, pump, or in pump pocket.
Ventricular Arrhythmias
- While some patients may transiently tolerate ventricular arrhythmias, prolonged ventricular arrhythmias can precipitate RV failure leading to decreased LV preload, low flows, and suction events
- Precipitating factors: Ischemia, electrolyte derangements, diseased myocardium with scar substrate, or LVAD specific etiologies such as suction event or malpositioned inflow cannula
Special Considerations for ventricular arrhythmias in setting of LVAD
-
- Treat reversible causes: correct electrolyte derangements, PCI for ischemia (RV MI), correct etiology of suction event (hypovolemia/RV failure)
- If unstable, perform synchronized cardioversion or defibrillation as appropriate (pads front/back to avoid placing directly over device)
- Utilize antiarrhythmic agents such as amiodarone/lidocaine
Use Anterior/Posterior Pad Placement for Shocks

Aortic Regurgitation
Okay wait, there are already too many things I need to look for! Why should I care about aortic regurgitation as a front-line provider?
- First, it is common, incidence of 6-32% at 1 year and 24-33% at 3 years
- Second, it is associated with increased hospitalizations and mortality
- The decreased aortic valve (AV) opening, associated with continuous flow LVADs, predisposes the leaflets to fusion/deterioration, which in addition to the reverse pressure gradient (low pressure in LV/high pressure in aorta) from the LVAD, can predispose these patients to develop AR
- Third, severe AR causes the LVAD to be ineffective which may result in cardiogenic shock, pulmonary edema, and RV failure despite elevated LVAD flows
Special Considerations for AR in setting of LVAD
-
- Always suspect when there is evidence of low cardiac output despite elevated LVAD flows (controller: elevated flow/power, decreased PI)
- As with all regurgitant lesions, consider afterload reduction, diuresis if volume overloaded (see CV-EMCrit-Acute Valve Disasters), may need speed adjustment
- CONSULT early, may require MCS for stabilization (if BTT) prior to surgical/ transcatheter valve replacement or transplant
PMID: 34980564, PMID: 32473380
Step 5: Transfer in Coordination with LVAD Coordinator
Finally, the step you have all been waiting for: TRANSFER 😂
It is imperative that LVAD patients that require admission are transferred to an LVAD center as they require expertise and resources that are likely not available at non-LVAD centers.
***Management of LVAD Patients in Extremis***
As with every patient in extremis, start with assessing the ABCs including level of consciousness. Simultaneously delegate someone to call the LVAD team.
Airway and Breathing can be approached similarly to any other patient in extremis, remembering that pulse oximetry will likely be completely unreliable without pulsatile flow. Circulation can be more challenging as many of these patients will not have a pulse at baseline and establishing a RTF (MAP) can be challenging even on a good day.
Extremely LOW threshold to place arterial line.
- Enables most accurate RTF (MAP) measurement. Crash femoral arterial line can be established quickly
- Access for labs (chemistry/ABG) to guide resuscitation
- Can assist with assessing if CPR is indicated and useful to monitoring quality of CPR
Patient ABCs, then VAD ABCs…
See this fantastic emergency guide (HM3 section) for step by step guide to change LVAD batteries, controller, and tighten driveline. This practical guide can be LIFESAVING.

Identify and treat reversible causes of hemodynamic instability placing patient in extremis. Perform cardioversion (chemical or electrical) and defibrillation as indicated based on perfusion status.
In the event of LVAD failure, be prepared to treat patients for cardiogenic shock
with inotropes and vasopressors as many of these patients have very poor intrinsic cardiac function. Norepinephrine is generally considered first line in CS as epinephrine has been associated with increased arrhythmias, myocardial oxygen consumption, and refractory shock; however, there is an inherent selection bias in many of these studies as epinephrine is often used in patients that are refractory to initial inopressor therapies thus introducing confounding.
Low dose epinephrine has been shown to be beneficial by increasing contractility and coronary perfusion with minimal effects on metabolism and is commonly used at low doses as an inopressor in CS. Dobutamine or milrinone can also be considered for inotropy with a recent study (DOREMI) showing no significant difference in outcomes; however, practically dobutamine with its shorter half-life and easier accessibility in many settings make it an ideal choice for emergency settings. [PMID: 35925990, PMID: 21037469, PMID: 29976291, PMID: 33797431, PMID: 13698421]
If the LVAD is still non-functional after trouble shooting and the patient is not perfusing it is time to initiate CPR. Despite the initial concern of LVAD dislodgement, studies have shown that standard chest compressions appear to be safe with no evidence of cannula dislodgement. [PMID: 24472494]

Special considerations for ACLS in LVAD patients
- Replace pulse checks with “perfusion checks,” using MAP equivalent and change in ETCO2
- Perform CPR and defibrillation (pads front/back to avoid LVAD) per standard ACLS protocols
- If the patient is Bridge-to-transplant (BTT), rapid initiation of venoarterial ECMO (VA ECMO) should be considered if available at your institution
- Continue trouble shooting LVAD with LVAD team assistance
Take Home messages:
- Contact the patient’s LVAD coordinator or nearest LVAD center EARLY
- For patients presenting in extremis—always trouble shoot the LVAD first
- LVAD patients in cardiac arrest with a non-functional LVAD should be treated with standard ACLS including CPR and defibrillation
- Consider ECMO in LVAD patients in extremis who are bridge to transplant
Additional Reading:
- SIMPLIFIED ALGORITHM FOR ACLS IN LVAD PATIENTS, CRITICAL CARE NOW
- EMS guide on LVAD management (section on HM3)
- Physiology and Clinical Utility of HeartMate Pump Parameters
- complications of LVADS
Additional New Information
More on EMCrit
Left Ventricular Assist Devices (LVADS)
Additional Resources
Awake & Aware Physicians Conference
January 13-15th, 2023 | Sedona, AZ
Check out the Conference Here
- EMCrit 373 – Mike Weinstock with another Critical Care Bounceback: “Asymptomatic Hypertension” - April 18, 2024
- EMCrit Wee – Ross Prager on 10 Heuristics for the New ICU Attending - April 13, 2024
- EMCrit 372 – FoundStab Intubation SOP - April 5, 2024
















This is such an amazing Article to read
am really happy to see this!!
Fantastic juicy post! Thanks Doc Katrina
Amazing podcast and a great post. Thank you for the good stuff.